Submitted to
Public Health Agency of Canada
POR-ROP@hc-sc.gc.ca
Prepared By
Leger
Ce rapport est aussi disponible en français.
POR 104-16
Contract Number 6D034-164442/001/CY
Awarded 2017-01-30
Project 15181-003
2017-03-31
Leger
507 Place d'Armes, Suite 700
Montréal, Quebec
H2Y 2W8
Phone: 514-982-2464
Fax: 514-987-1960
www.leger360.com
Leger is pleased to present the Public Health Agency of Canada with this report on findings from a quantitative survey aimed to evaluate seasonal influenza immunization coverage within the Canadian population.
This report was prepared by Leger who was contracted by the Public Health Agency of Canada (contract number 6D034-164442/001/CY awarded January 30, 2017).
The main objective of the study is to evaluate seasonal influenza immunization coverage within the Canadian population. The following topics are covered among Canadian households participating in the survey.
The quantitative research was conducted by telephone, using Computer Assisted Telephone Interviewing (CATI) technology. This approach is the most appropriate to assess seasonal influenza immunization coverage among different subgroups of the Canadian population, while ensuring a high level of representativeness. To achieve data reliability in all subgroups, a sample of 2,024 adult Canadians were surveyed. Only one adult respondent was surveyed within each household.
Fieldwork for the survey was conducted from February 14 to March 5, 2017. The national response rate for the survey was 20.3%. Complete call dispositions are presented in Appendix A. A pre-test of 30 interviews, 15 in each official language, was completed on February 14, 2017 (in English) and on February 16, 2017 (in French). Survey interviews lasted 7 minutes on average.
Telephone interviewing was conducted using Leger's Computer-Assisted Telephone Interviewing (CATI) technology. Leger's CATI system handles sampling electronically, randomly selecting and dialing the phone number to call. To ensure perfect coverage of a population, the sample consisted of residential phone numbers located in all Canadian provinces and territories, as well as cell phone numbers of Canadians who do not have a residential landline phone number (i.e. pre-validated cell-only numbers). Based on the Communications Monitoring Report 2016 published by the CRTC in 2016, which revealed that 23.7% of Canadian households were using a cell phone exclusively, Leger made sure that 23.7% of the final sample was derived from cell-only numbers.
Landline phone numbers were generated and cell-only numbers were purchased according to a stratified regional sampling approach. Regional quotas were applied to ensure that a sufficient number of interviews within each region of Canada and within each type of phone number (residential (1,527) and cell phone (497)). In addition to these regional quotas, fieldwork was conducted to ensure a good distribution of respondents in terms of gender (men and women) and language (English and French-speaking Canadians), using soft quotas.
A total of 2,024 adult Canadians were interviewed in all regions of the country. The national margin of error for the survey is +/- 2.18%, 19 times out of 20.
Based on data from Statistics Canada's 2011 national census, Leger weighted the results of this survey by age, gender, region and language (mother tongue). Results were also weighted based on the presence of minor children in the household (yes or no), since this question was asked and is important for this study. Moreover, the weight of respondents reached via the cell-only sample was also controlled to match the expected 23.7% after weighting.
As a Certified Gold-Seal MRIA Member, Leger adheres to the most stringent guidelines for quantitative research. The survey has been registered with the MRIA in accordance with Government of Canada requirements for quantitative research, including the MRIA Code of Conduct and Standards of the Conduct of Government of Canada Public Opinion Research - Series D - Quantitative Research.
Respondents were assured of the voluntary and confidentiality aspect of the approach and of the anonymity of their responses. As with all research conducted by Léger, all information that could allow for the identification of participants was removed from the data, in accordance with the Privacy Act of Canada.
The details of the methodology procedure and more information on Leger's quality control mechanisms are presented in Appendix A.
The French and English questionnaires (the same questionnaire was used for both languages of the study) are available in Appendix B.
Details regarding the weighting procedures can be found in Appendix A and detailed statistical tables of results can be found in Appendix C.
The views and observations expressed in this document do not reflect those of the Public Health Agency of Canada. This report was compiled by Leger based on the research conducted specifically for this project. This research is probabilistic; the results can be inferred to the general population of Canada. The design of the research was built with this objective in mind.
Leger certifies that the final deliverables fully comply with the Government of Canada's political neutrality requirements outlined in the Communications Policy of the Government of Canada and Procedures for Planning and Contracting Public Opinion Research.
Supplier name: Leger
PWGSC Contract Number: 6D034-164442/001/CY
Contract Award Date: January 30th, 2017
The expenditure for this project is $74,664.75 (including HST).
To obtain more information on this study, please email POR-ROP@hc-sc.gc.ca
This chapter presents the detailed findings from the quantitative survey for the Public Health Agency of Canada regarding seasonal influenza immunization coverage within the Canadian population. The research was fielded between February 14 and March 5, 2017. A full set of detailed tables is also available in the appendix (under a separate cover).
In this report, it should be noted that the numbers presented have been rounded. On the other hand, numbers before rounding have been used to calculate the sums presented. For this reason, those sums may not match the manual summation of the numbers presented.
According to the normal distribution, a two-tailed test is always done between two proportions and based on the unweighted total columns. The test is performed by comparing the percentage of a vertical cell with the vertical percentage formed by the complement of the cells for the relevant category (e.g. the complement of men is women and the complement of the 18-24 age group is those 25 and over). The test results (if they are significant at a confidence level of 95%) are indicated with a symbol in the table.
In the tables, results presented with a "↓" characters represent statistically lower differences when compared to the complement, while results with "↑" characters indicate statistically higher differences when compared to the complement.
Nearly six out of ten Canadians (59%) said that they had received the seasonal flu vaccine before September 1, 2016. When looking at subgroups within the general population, we observe that people 65+ years old (79%), people from the Maritimes (72%), the 18-64 group with a chronic condition (68%), those with a university degree (66%), Ontarians (64%) and females (64%) are more likely to have received the seasonal flu vaccine in the past.
| Total | Region | ||||||
|---|---|---|---|---|---|---|---|
| Maritimes | Quebec | Ontario | MB/SK/AB | BC | Territories | ||
| Weighted n= | 2,024 | 141 | 479 | 777 | 355 | 266 | 6 |
| Unweighted n= | 2,024 | 160 | 526 | 640 | 388 | 280 | 30 |
| Yes | 59% | 72%↑ | 48%↓ | 64%↑ | 59% | 59% | 58% |
| No | 41% | 28%↓ | 52%↑ | 36%↓ | 41% | 41% | 42% |
Since September 1, 2016, more than a third of the Canadian population (36%) received the seasonal influenza vaccine. We can observe that those 65 years of age or older (69%) and those between the ages of 18 and 64 with a chronic condition (37%) are more likely to have received the flu vaccine this year. We also note that respondents from the Maritimes (50%) and women (39%) are more likely to have received the flu vaccine since September. The opposite trend is observed in Quebec, where one-quarter of respondents (25%) received the flu vaccine this year.
| Total | Subgroups | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| 18-64 | 18-64 with chronic health condition | 18-64 without chronic health condition | 65 + | Children 13-17 years of age | Children 6-12 years of age | Children 6 months to 5 years of age | Children younger than 6 months | ||
|
Table 1 footnotes
| |||||||||
| Weighted n= | 2,024 | 1,647 | 412 | 1,235 | 377 | 213 | 309 | 254 | 20 |
| Unweighted n= | 2,024 | 1,446 | 407 | 1,039 | 578 | 185 | 255 | 199 | 18Table 1 footnote * |
| Yes | 36% | 28%↓ | 37%↑ | 25%↓ | 69%↑ | 27% | 21%↓ | 22%↓ | 20% |
| No | 64% | 72%↑ | 63%↓ | 75% ↑ | 31%↓ | 73% | 79%↑ | 78%↑ | 80% |
| Total | Region | Gender | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Maritimes | Quebec | Ontario | MB/SK/AB | BC | Territories | Male | Female | ||
| Weighted n= | 2,024 | 141 | 479 | 777 | 355 | 266 | 6 | 980 | 1,044 |
| Unweighted n= | 2,024 | 160 | 526 | 640 | 388 | 280 | 30 | 801 | 1,223 |
| Yes | 36% | 50%↑ | 26%↓ | 37% | 38% | 38% | 39% | 32%↓ | 39%↑ |
| No | 64% | 50%↓ | 74%↑ | 63% | 62% | 62% | 61% | 68%↑ | 61%↓ |
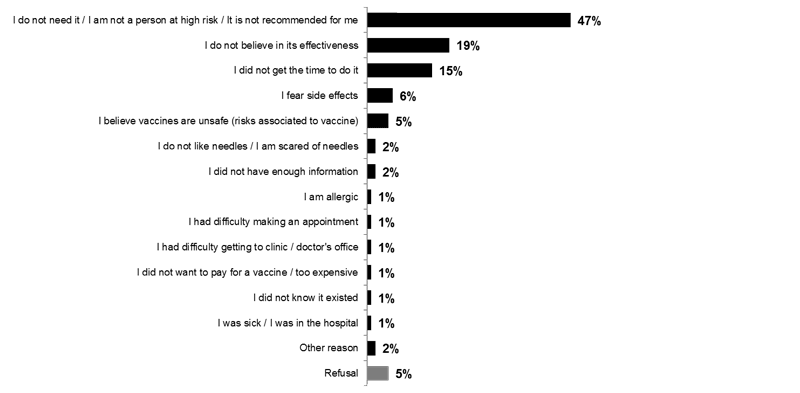
Nearly half of Canadians who indicated that they did not receive the flu vaccine this year (47%) say that they did not get the flu vaccine because they did not need it / they are not a person at risk / it is not recommended for them. Two Canadians out of ten (19%) say that they do not believe in the effectiveness of the vaccine and 15 percent say they did not have time to do it.
What are the reasons why you did not receive the flu vaccine this year?
Base: Among those who did not receive the flu vaccine this year (n=1,171)

Because respondents were able to give multiple answers, total mentions may exceed 100%.
Conversely, Table 3 shows that 44 percent of those who received the flu vaccine this year did so because they want to prevent infection / do not want to get sick. This reason is especially prevalent among the 65+ group (51%). To a lesser extent, 16 percent of those who received the vaccine did so because it is required in their workplace and 13 percent did so because they are at risk because of their health condition or because they receive it every year.
The 18-64 group without a chronic condition is more likely to have received the flu vaccine because it is required in their workplace (25%) while those in the 18-64 group with a chronic condition are more likely to say that they got the flu vaccine because of their risky health condition (31%). Finally, respondents in the 65+ group are showing a higher proportion of getting the flu vaccine because they receive it every year (22%) or because it was recommended by a health care professional (12%).
| Base: Among those who received the flu vaccine this year (n=853) | Total | Subgroups | |||
|---|---|---|---|---|---|
| 18-64 | 18-64 with chronic health condition | 18-64 without chronic health condition | 65 + | ||
| Weighted n= | 724 | 462 | 152 | 310 | 262 |
| Unweighted n= | 853 | 450 | 166 | 284 | 403 |
| I want to prevent infection / I do not want to get sick | 44% | 41% | 35% | 44% | 51%↑ |
| It is required in my workplace | 16% | 23% | 18% | 25%↑ | 4%↓ |
| I am at risk because of my health condition | 13% | 13% | 31%↑ | 5%↓ | 12% |
| I receive it every year (no specific reason) | 13% | 8%↓ | 11% | 7%↓ | 22%↑ |
| I was recommended by a health care professional | 9% | 7% | 9% | 6% | 12%↑ |
| If not vaccinated, I can transmit the disease to at-risk people (children, elderly or sick people/patients) | 7% | 8% | 7% | 9% | 4%↓ |
| I am at risk because of my age | 6% | 2% | 4% | 2%↓ | 11%↑ |
| If not vaccinated, I can transmit the disease to family members, colleagues or friends (without mention of at-risk people) | 5% | 7% | 5% | 9% | 1%↓ |
| I was encouraged by family members, colleagues or friends | 3% | 4% | 2% | 4% | 1%↓ |
| It was offered / free (by employer or other) | 2% | 2% | 1% | 2% | 2% |
| I would be required to wear a mask at work if not vaccinated | 0% | 0% | 0% | 0% | 0% |
| Other reason | 0% | 0% | 0% | 0% | 0% |
| Refusal | 1% | 1% | 0% | 1% | 0% |
Because respondents were able to give multiple answers, total mentions may exceed 100%.
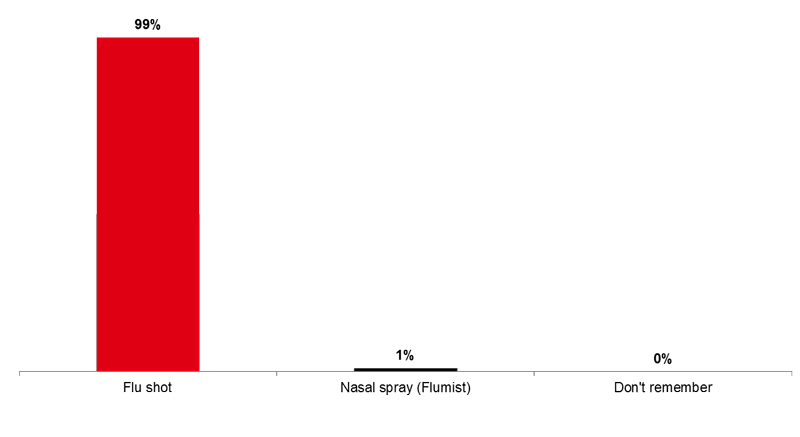
Nearly all Canadians who received the flu vaccine this year (99%) were vaccinated with a flu shot (with a needle), whereas one percent received it by nasal spray (Flumist).
Image 2. Form of Flu Vaccine Received (Q4)
Was the flu vaccine you received this year given by a shot (with a needle) or a spray in the nose?
Base: Among those who received the flu vaccine this year (n=853)
Most Canadians who received the flu vaccine this year received it in October 2016 (38%) or in November 2016 (37%). Table 4 shows that two-thirds of Quebecers (67%) received the vaccine in November and half of residents of Manitoba, Saskatchewan or Alberta (51%) received it in October. The analysis of the results shows that there are no significant differences between the subgroups studied by Public Health Agency of Canada (PHAC).
Base: Among those who received the flu vaccine this year (n=853) |
Total | Region | |||||
|---|---|---|---|---|---|---|---|
| Maritimes | Quebec | Ontario | MB/SK/AB | BC | Territories | ||
|
Table 4 footnotes
|
|||||||
| Weighted n= | 724 | 71 | 127 | 290 | 134 | 101 | 2 |
| Unweighted n= | 853 | 86 | 166 | 288 | 176 | 125 | 12Table 4 footnote * |
| September 2016 | 4% | 5% | 3% | 5% | 5% | 2% | 7% |
| October 2016 | 38% | 40% | 15%↓ | 40% | 51%↑ | 39% | 8% |
| November 2016 | 37% | 28% | 67%↑ | 32%↓ | 26%↓ | 35% | 46% |
| December 2016 | 9% | 9% | 10% | 9% | 5%↓ | 10% | 18% |
| January 2017 | 4% | 5% | 3% | 4% | 5% | 4% | 0% |
| February 2017 | 1% | 0% | 0% | 1% | 2% | 1% | 0% |
| Don't remember | 8% | 13%↑ | 3%↓ | 8% | 6% | 9% | 21% |
One-third of Canadians who got the flu vaccine (33%) received it at a doctor's office or health clinic. This proportion is higher among respondents from the Maritimes (43%) and Ontario (45%).
More than one-quarter of respondents (28%) received the flu vaccine at their pharmacy. This was especially prominent in British Columbia (36%).
Finally, Quebec has the highest proportion of respondents who received the flu vaccine at a temporary vaccine clinic (22%) or at a CLSC or community health centre (26%).
| Base: Among those who received the flu vaccine this year (n=853) | Total | Region | |||||
|---|---|---|---|---|---|---|---|
| Maritimes | Quebec | Ontario | MB/SK/AB | BC | Territories | ||
|
Table 5 footnotes
| |||||||
| Weighted n= | 724 | 71 | 127 | 290 | 134 | 101 | 2 |
| Unweighted n= | 853 | 86 | 166 | 288 | 176 | 125 | 12Table 5 footnote * |
| Doctor's office or health clinic | 33% | 43%↑ | 14%↓ | 45%↑ | 21%↓ | 29% | 27% |
| Pharmacy | 28% | 28% | 9%↓ | 31% | 34% | 36%↑ | 0% |
| Temporary vaccine clinic (i.e. at the mall) | 11% | 10% | 22%↑ | 7%↓ | 17%↑ | 4%↓ | 7% |
| CLSC or Community health centre | 10% | 3%↓ | 26%↑ | 5%↓ | 13% | 6% | 51% |
| Workplace | 9% | 11% | 12% | 4%↓ | 8% | 19%↑ | 16% |
| Hospital | 7% | 3% | 9% | 8% | 5% | 4% | 0% |
| Retirement residence or eldercare centre | 1% | 0% | 3%↑ | 0% | 1% | 2% | 0% |
| Other | 1% | 0% | 5%↑ | 0% | 0% | 1% | 0% |
| Don't remember | 0% | 0% | 1%↑ | 0% | 0% | 0% | 0% |
Of the households with children, nearly one-quarter of Canadian children (23%) received the flu vaccine this year. Interestingly, influenza immunization coverage seems to decrease when there are more children in the household.
| Total | Number of children | ||||
|---|---|---|---|---|---|
| 1 child | 2 children | 3 children | 4+ children | ||
| Weighted n= | 1.081 | 233 | 468 | 225 | 154 |
| Unweighted n= | 894 | 204 | 380 | 174 | 136 |
| Yes | 23% | 31% | 23% | 19% | 16% |
| No | 77% | 69% | 77% | 81% | 84% |
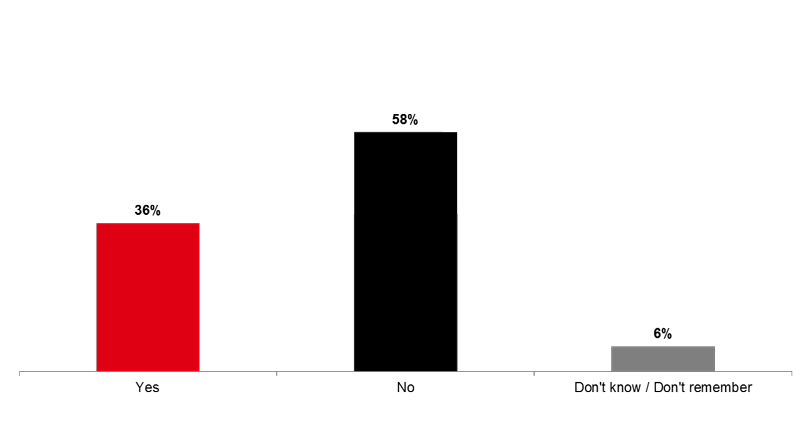
For more than a third of the children (36%), it was the first time they received the flu vaccine.
Image 3. First Time Receiving the Flu Vaccine (Q12)
Was it the first time your ***<Q9A1> <Q9A2>***-old child received the flu vaccine?
Base: Among all children who received the flu vaccine (n=124)
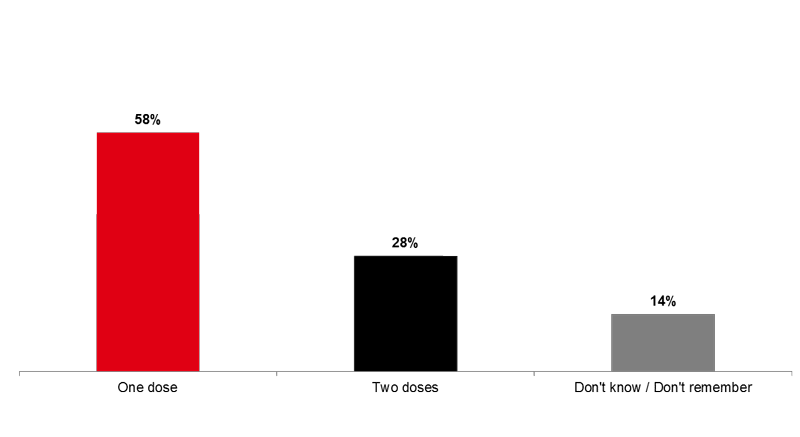
Nearly six out of ten of all children between 6 months and 9 years of age who received the flu vaccine (58%) received only one dose, while 28 percent received the two recommended doses.
Image 4. Number of doses received by children between 6 months and 9 years of age, receiving the vaccine for the first time (Q13)
Since September 1st, 2016 inclusively, has your ***<Q9A1> <Q9A2>***-old child received one or two doses of the flu vaccine?
Base: Among all children between 6 months and 9 years of age who received the flu vaccine for the first time (n=40)
Nearly two in ten parents indicated that their child(ren) did not receive a second dose of the flu vaccine because they did not have this information (18%) and they did not know there was a second dose (16%).
Image 5. Reasons for Children Not Receiving a Second Dose (Q14)
Children between the ages of 6 months and 9 years who are receiving the flu vaccine for the first time are sometimes given two doses. What are the reasons why your ***<Q9A1> <Q9A2>***-old child did not receive a second vaccine?
Base: Among children between 6 months and 9 years of age who did not receive a second dose (n=26Image 5 footnote *)
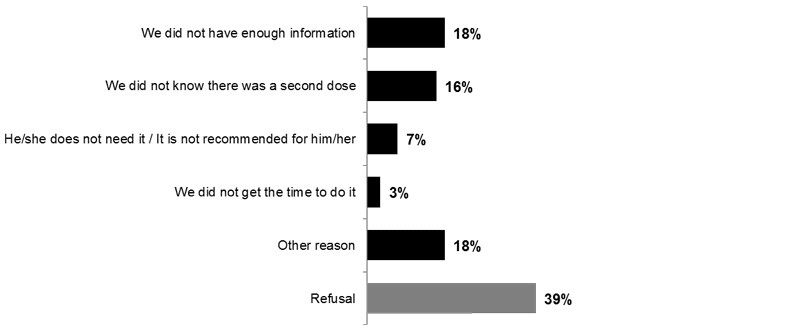
Image 5 footnotes
Because of the small sample size (n<30), results are presented for illustrative purposes only.
Note: Only answers with at least 1% of total mentions are presented.
Because respondents were able to give multiple answers, total mentions may exceed 100%.
Of those who have children under 18 years of age, the main reasons provided for not getting their children vaccinated are the belief that they don't need it (36%) or not believing in its effectiveness (24%).
Image 6. Reasons for Children Not Receiving the Vaccine This Year (Q15)
What is(are) the reason(s) your ***<Q9A1> <Q9A2>***-old child did not receive the flu vaccine this year?
Base: Among all children who did not receive the flu vaccine (n=739)
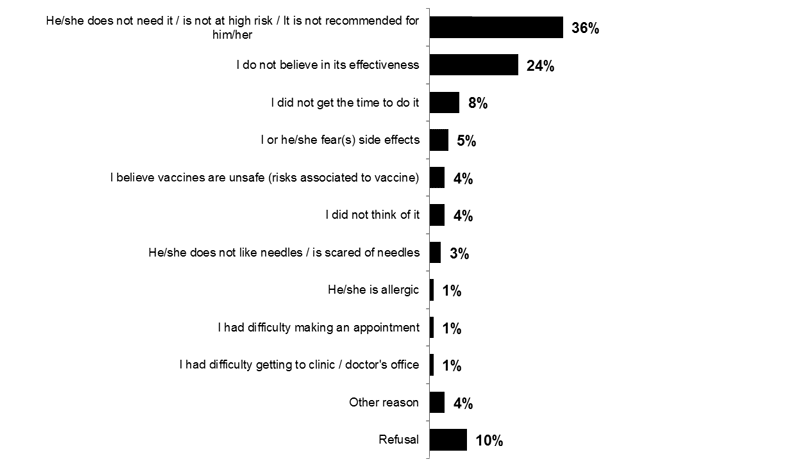
Note to reader: Only answers with at least 1% of total mentions are presented.
Because respondents were able to give multiple answers, total mentions may exceed 100%.
Two out of ten respondents (22%) say that they had the flu within the past 12 months, mostly with mild symptoms (19%). It seems that respondents from Manitoba, Saskatchewan and Alberta (17%) are slightly less likely to suffer from flu symptoms.
| PERSONALLY, WITHIN THE PAST 12 MONTHS | Total | Region | |||||
|---|---|---|---|---|---|---|---|
| Maritimes | Quebec | Ontario | MB/SK/AB | BC | Territories | ||
| Weighted n= | 2,024 | 141 | 479 | 777 | 355 | 266 | 6 |
| Unweighted n= | 2,024 | 160 | 526 | 640 | 388 | 280 | 30 |
| NET YES | 22% | 28% | 25% | 21% | 17%↓ | 25% | 46%↑ |
| Yes, with severe symptoms (e.g. hospitalization, pneumonia) | 3% | 4% | 2%↓ | 3% | 4% | 5% | 5% |
| Yes, with mild symptoms (e.g. sudden onset of high fever, chills, sore throat, cough, muscle pain) | 19% | 24% | 23%↑ | 18% | 14%↓ | 20% | 41%↑ |
| No, did not have the flu | 77% | 72% | 74% | 78% | 82%↑ | 73% | 54%↓ |
| Don't know / Don't remember | 1% | 0% | 1% | 1% | 1% | 2% | 0% |
Conversely, as shown in Table 7b, the 18-64 group with a chronic health condition and children 6 months to 5 years of age are more likely to have shown flu symptoms in the past year (29% and 32%, respectively). Regardless of whether they were vaccinated or not, the same proportion of Canadians was affected by flu symptoms within the last months.
| PERSONALLY, WITHIN THE PAST 12 MONTHS | Total | Subgroups | Flu vaccine 2016-2017 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 18-64 | 18-64 with chronic health condition | 18-64 without chronic health condition | 65 + | Children 13-17 years of age | Children 6-12 years of age | Children 6 months to 5 years of age | Children younger than 6 months | Yes | No | ||
|
Table 7 footnotes
| |||||||||||
| Weighted n= | 2,024 | 1,647 | 412 | 1,235 | 377 | 213 | 309 | 254 | 20 | 724 | 1,300 |
| Unweighted n= | 2,024 | 1,446 | 407 | 1,039 | 578 | 185 | 255 | 199 | 18Table 7b footnote * | 853 | 1,171 |
| NET YES | 22% | 24% | 29%↑ | 22% | 16%↓ | 22% | 22% | 32%↑ | 19% | 22% | 23% |
| Yes, with severe symptoms (e.g. hospitalization, pneumonia) | 3% | 3% | 7%↑ | 2%↓ | 3% | 3% | 1% | 5% | 6% | 4% | 3% |
| Yes, with mild symptoms (e.g. sudden onset of high fever, chills, sore throat, cough, muscle pain) | 19% | 20% | 22% | 20% | 13%↓ | 19% | 21% | 27%↑ | 13% | 18% | 20% |
| No, did not have the flu | 77% | 75% | 71%↓ | 77% | 83%↑ | 76% | 77% | 65%↓ | 81% | 77% | 76% |
| Don't know / Don't remember | 1% | 1% | 1% | 1% | 1% | 2% | 1% | 3%↑ | 0% | 1% | 1% |
It seems that respondents have seen more than half (56%) of their friends or family members suffer from severe (13%) and/or mild (47%) flu symptoms. Friends or family members from the Maritimes (20%) and British Columbia (18%) are more likely to have more severe flu symptoms.
| FRIEND OR FAMILY MEMBER, WITHIN THE PAST 12 MONTHS | Total | Region | |||||
|---|---|---|---|---|---|---|---|
| Maritimes | Quebec | Ontario | MB/SK/AB | BC | Territories | ||
| Weighted n= | 2,024 | 141 | 479 | 777 | 355 | 266 | 6 |
| Unweighted n= | 2,024 | 160 | 526 | 640 | 388 | 280 | 30 |
| NET YES | 56% | 59% | 54% | 56% | 56% | 61% | 53% |
| Yes, with severe symptoms (e.g. hospitalization, pneumonia) | 13% | 20%↑ | 9%↓ | 13% | 14% | 18%↑ | 8% |
| Yes, with mild symptoms (e.g. sudden onset of high fever, chills, sore throat, cough, muscle pain) | 47% | 49% | 46% | 47% | 46% | 49% | 50% |
| No, did not have the flu | 40% | 36% | 42% | 41% | 41% | 37% | 38% |
| Don't know / Don't remember | 3% | 5% | 4% | 3% | 3% | 2% | 9% |
Because respondents were able to give multiple answers, total mentions may exceed 100%.
More than half of respondents (55%) mentioned having severe (7%) or mild (48%) flu symptoms in the past. Respondents from the Maritimes (14%) and British Columbia (11%) are slightly more likely to have experienced severe flu symptoms in the past.
| PERSONALLY, BEFORE THE PAST 12 MONTHS |
Total | Region | |||||
|---|---|---|---|---|---|---|---|
| Maritimes | Quebec | Ontario | MB/SK/AB | BC | Territories | ||
| Weighted n= | 2,024 | 141 | 479 | 777 | 355 | 266 | 6 |
| Unweighted n= | 2,024 | 160 | 526 | 640 | 388 | 280 | 30 |
| NET YES | 55% | 62% | 53% | 55% | 57% | 57% | 45% |
| Yes, with severe symptoms (e.g. hospitalization, pneumonia) | 7% | 14%↑ | 3%↓ | 6% | 9% | 11%↑ | 13% |
| Yes, with mild symptoms (e.g. sudden onset of high fever, chills, sore throat, cough, muscle pain) | 48% | 49% | 49% | 48% | 48% | 45% | 33% |
| No, did not have the flu | 41% | 35% | 45%↑ | 42% | 41% | 37% | 47% |
| Don't know / Don't remember | 3% | 3% | 2% | 3% | 2% | 6%↑ | 7% |
Because respondents were able to give multiple answers, total mentions may exceed 100%.
Table 9b shows that children between 13-17 years of age (12%) and those who received the vaccine this year (11%) are slightly more likely to have experienced severe flu symptoms in the past.
| PERSONALLY, BEFORE THE PAST 12 MONTHS | Total | Subgroups | Flu vaccine 2016-2017 | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 18-64 | 18-64 with chronic health condition | 18-64 without chronic health condition | 65 + | Children 13-17 years of age | Children 6-12 years of age | Children 6 months to 5 years of age | Children younger than 6 months | Yes | No | ||
|
Table 9b footnotes
| |||||||||||
| Weighted n= | 2,024 | 1,647 | 412 | 1,235 | 377 | 213 | 309 | 254 | 20 | 724 | 1,300 |
| Unweighted n= | 2,024 | 1,446 | 407 | 1,039 | 578 | 185 | 255 | 199 | 18Table 9b footnote * | 853 | 1,171 |
| NET YES | 55% | 57% | 60% | 56% | 49%↓ | 58% | 60% | 67%↑ | 52% | 56% | 55% |
| Yes, with severe symptoms (e.g. hospitalization, pneumonia) | 7% | 7% | 9% | 6%↓ | 8% | 12%↑ | 8% | 10% | 5% | 11%↑ | 5% ↓ |
| Yes, with mild symptoms (e.g. sudden onset of high fever, chills, sore throat, cough, muscle pain) | 48% | 50% | 51% | 50% | 41%↓ | 46% | 52% | 56%↑ | 47% | 45%↓ | 50%↑ |
| No, did not have the flu | 41% | 40% | 36% | 41% | 48%↑ | 41% | 37% | 29%↓ | 48% | 42% | 41% |
| Don't know / Don't remember | 3% | 3% | 3% | 3% | 3% | 1% | 3% | 4% | 0% | 3% | 4% |
More than six out of ten respondents (63%) are aware of friends or family members who have suffered from severe (16%) and/or mild (55%) flu symptoms in the past.
Respondents' friends or family members in the Maritimes (73%) are more likely to have suffered flu symptoms.
| FRIEND OR FAMILY MEMBER, BEFORE THE PAST 12 MONTHS | Total | Region | |||||
|---|---|---|---|---|---|---|---|
| Maritimes | Quebec | Ontario | MB/SK/AB | BC | Territories | ||
| Weighted n= | 2,024 | 141 | 479 | 777 | 355 | 266 | 6 |
| Unweighted n= | 2,024 | 160 | 526 | 640 | 388 | 280 | 30 |
| NET YES | 63% | 73%↑ | 55%↓ | 66% | 64% | 66% | 65% |
| Yes, with severe symptoms (e.g. hospitalization, pneumonia) | 16% | 24%↑ | 8%↓ | 17% | 15% | 22%↑ | 25% |
| Yes, with mild symptoms (e.g. sudden onset of high fever, chills, sore throat, cough, muscle pain) | 55% | 63%↑ | 47%↓ | 56% | 57% | 57% | 59% |
| No, did not have the flu | 30% | 22%↓ | 39%↑ | 27% | 29% | 28% | 26% |
| Don't know / Don't remember | 6% | 5% | 6% | 7% | 7% | 6% | 10% |
Because respondents were able to give multiple answers, total mentions may exceed 100%.
The vast majority of respondents (93%) have seen a health care provider in the last year. Three-quarters of Canadians (76%) have seen their family doctor. This proportion is slightly higher among respondents from Manitoba, Saskatchewan and Alberta (81%).
Nearly two-thirds of respondents (64%) visited their dentist (orthodontist) in the last 12 months. This is especially prominent in Ontario (69%).
Close to six out of ten Canadians (58%) have seen their pharmacist in the last year, a proportion that is higher in the Maritimes (66%). Finally, more than a third of respondents (37%) visited a medical specialist within the last 12 months and 22 percent have seen a nurse.
| Total | Region | ||||||
|---|---|---|---|---|---|---|---|
| Maritimes | Quebec | Ontario | MB/SK/AB | BC | Territories | ||
| Weighted n= | 2,024 | 141 | 479 | 777 | 355 | 266 | 6 |
| Unweighted n= | 2,024 | 160 | 526 | 640 | 388 | 280 | 30 |
| NET seen a health care provider | 93% | 95% | 92% | 93% | 92% | 94% | 82%↓ |
| Family doctor | 76% | 81% | 68%↓ | 77% | 81% ↑ | 78% | 63% |
| Dentist / orthodontist | 64% | 71% | 56%↓ | 69%↑ | 60% | 62% | 52% |
| Pharmacist | 58% | 66%↑ | 54% | 59% | 56% | 57% | 47% |
| Medical specialist (e.g. gynecologist, cardiologist, ophthalmologist, allergist, orthopedist, psychiatrist) | 37% | 33% | 37% | 41%↑ | 34% | 30%↓ | 33% |
| Nurse practitioner | 22% | 21% | 28%↑ | 23% | 15%↓ | 15%↓ | 38%↑ |
| Midwife | 1% | 1% | 1% | 1% | 1% | 1% | 3% |
| Did not see a health care provider | 7% | 5% | 8% | 7% | 8% | 6% | 18%↑ |
| Refusal | 0% | 0% | 0% | 0% | 0% | 0% | 0% |
Because respondents were able to give multiple answers, total mentions may exceed 100%.
In the last year, nearly one-quarter of respondents (23%) have seen an alternative care provider from the list provided, chiropractors (17%) being the most popular.
Table 12 shows that Quebecers (84%) are more likely not to have seen any alternative care providers in the last 12 months.
| Total | Region | ||||||
|---|---|---|---|---|---|---|---|
| Maritimes | Quebec | Ontario | MB/SK/AB | BC | Territories | ||
| Weighted n= | 2,024 | 141 | 479 | 777 | 355 | 266 | 6 |
| Unweighted n= | 2,024 | 160 | 526 | 640 | 388 | 280 | 30 |
| NET seen an alternative care provider | 23% | 18% | 16%↓ | 27%↑ | 23% | 27% | 12% |
| Chiropractor | 17% | 11%↓ | 12%↓ | 19% | 18% | 19% | 6% |
| Acupuncturist | 5% | 2% | 2%↓ | 7%↑ | 6% | 6% | 3% |
| Homeopath or naturopath | 4% | 5% | 4% | 5%↑ | 3% | 3% | 0% |
| Spiritual or religious healer | 2% | 1% | 0%↓ | 3%↑ | 1% | 2% | 3% |
| Herbalist | 1% | 0% | 1% | 1% | 1% | 1% | 6%↑ |
| Reflexologist | 1% | 1% | 1% | 2% | 0% | 2% | 0% |
| Did not see an alternative care provider | 76% | 82% | 84%↑ | 72%↓ | 77% | 73% | 88% |
| Refusal | 0% | 0% | 0% | 1%↑ | 0% | 1% | 0% |
Because respondents were able to give several answers, total mentions may exceed 100%.
Lastly, two out of ten respondents (22%) have seen other care providers in the last 12 months; including massage therapists (7%) and physiotherapists (6%).
Image 7. Other Care Providers Seen in the Last 12 Months (Q19OP)
Have you seen another health care provider in the past 12 months?
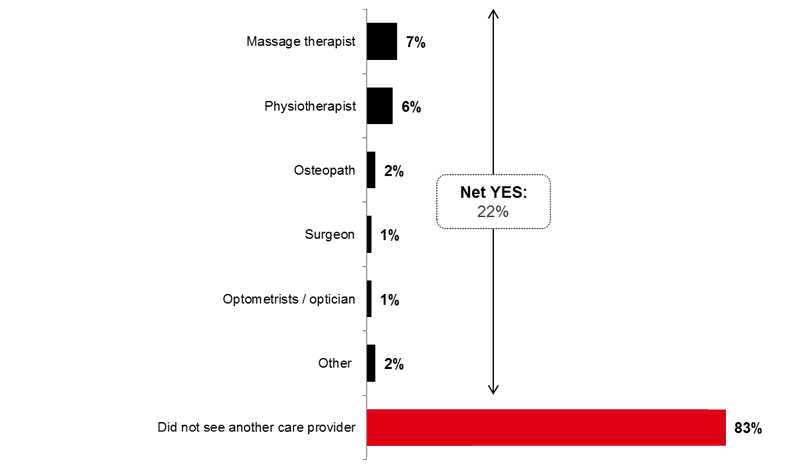
Because respondents were able to give multiple answers, total mentions may exceed 100%.
Nearly half of respondents (47%) state that when they want to know more about the flu shot, they look for information in a public health setting, health clinic or pharmacy. This proportion is higher among respondents from the Maritimes (60%) and among those 55 years of age or older (53%).
The Internet is also a source of information about the flu shot; 18 percent would do an online search and 13 percent would consult a government website. Quebecers (28%) are more likely to refer a government website to get information about the flu shot.
Those 18 to 34 years old are slightly more likely to seek information on a government website (18%) or by trusting word of mouth (11%) when looking for information about the flu shot.
| Total | Region | Age | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Maritimes | Quebec | Ontario | MB/SK/AB | BC | Territories | 18-34 | 35-54 | 55+ | ||
| Weighted n= | 2,024 | 141 | 479 | 777 | 355 | 266 | 6 | 564 | 748 | 711 |
| Unweighted n= | 2,024 | 160 | 526 | 640 | 388 | 280 | 30 | 355 | 655 | 1,014 |
| A public health setting, health clinic or pharmacy | 47% | 60%↑ | 41%↓ | 48% | 47% | 44% | 62% | 40%↓ | 45% | 53%↑ |
| Online search | 18% | 12%↓ | 15%↓ | 21%↑ | 17% | 22% | 22% | 20% | 20% | 14%↓ |
| A government website (e.g. Public Health Agency of Canada) | 13% | 4%↓ | 28%↑ | 10% | 8%↓ | 10%↓ | 14% | 18%↑ | 15% | 8%↓ |
| The media (e.g. newspaper, radio) | 11% | 9% | 7%↓ | 15%↑ | 11% | 11% | 6% | 6%↓ | 11% | 16%↑ |
| Word of mouth | 8% | 8% | 4%↓ | 11%↑ | 5%↓ | 11% | 0% | 11%↑ | 9% | 5%↓ |
| Social media (e.g. Twitter, Facebook) | 6% | 4% | 2%↓ | 8% | 8% | 9% ↑ | 0% | 8% | 8% | 4%↓ |
| Printed materials (e.g. pamphlets, books, medical journals) | 1% | 1% | 0%↓ | 2%↑ | 1% | 2% | 0% | 1% | 1% | 2% |
| An app (e.g. ImmunizeCA) | 0% | 1% | 0% | 0% | 0% | 0% | 0% | 1%↑ | 0% | 0%↓ |
| Other | 2% | 1% | 2% | 2% | 5%↑ | 1% | 3% | 3% | 2% | 2% |
| None of the above | 4% | 0%↓ | 5% | 5% | 3% | 2%↓ | 7% | 3% | 3% | 5%↑ |
| Does not apply to me | 11% | 14% | 11% | 9%↓ | 13% | 12% | 5% | 10% | 11% | 10% |
| Refusal | 0% | 0% | 0% | 0% | 1%↑ | 0% | 0% | 0% | 1% | 0% |
Because respondents were able to give multiple answers, total mentions may exceed 100%.
The most trusted resource about the flu shot out of the options provided is the family doctor. Nearly two-thirds of respondents (64%) say that they trust their family doctor the most for information about the flu shot. This proportion is higher in the Maritimes (72%) and Ontario (70%).
| Total | Region | Age | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Maritimes | Quebec | Ontario | MB/SK/AB | BC | Territories | 18-34 | 35-54 | 55+ | ||
| Weighted n= | 2,024 | 141 | 479 | 777 | 355 | 266 | 6 | 564 | 748 | 711 |
| Unweighted n= | 2,024 | 160 | 526 | 640 | 388 | 280 | 30 | 355 | 655 | 1,014 |
| Family doctor | 64% | 72%↑ | 57%↓ | 70%↑ | 58%↓ | 68% | 44%↓ | 58%↓ | 66% | 69%↑ |
| Pharmacist | 22% | 30%↑ | 24% | 21% | 20% | 21% | 8% | 19% | 22% | 24%↑ |
| Nurse practitioner | 14% | 16% | 18%↑ | 12% | 12% | 11% | 36%↑ | 15% | 13% | 13% |
| Medical specialist (e.g. gynecologist, cardiologist, ophthalmologist, allergist, orthopedist, psychiatrist) | 8% | 8% | 6%↓ | 10%↑ | 7% | 10% | 8% | 7% | 9% | 9% |
| Family | 6% | 3% | 5% | 9%↑ | 4%↓ | 6% | 0% | 11%↑ | 4%↓ | 5% |
| Friends | 4% | 4% | 3% | 5% | 4% | 1%↓ | 0% | 4% | 4% | 3%↓ |
| The government / Health Canada | 4% | 7% | 2%↓ | 4% | 7%↑ | 4% | 2% | 4% | 6%↑ | 3%↓ |
| Other health care provider (e.g. nurse, pharmacist, medical specialist) | 2% | 1% | 2% | 2% | 3% | 3% | 24%↑ | 3% | 2% | 2% |
| An alternative care provider (e.g. naturopath, homeopath) | 2% | 4% | 2% | 3% | 2% | 0% | 6% | 2% | 2% | 2% |
| I trust my own judgement | 2% | 3% | 2% | 2% | 3% | 2% | 3% | 2% | 3% | 3% |
| Government website | 1% | 0% | 0% | 1% | 2%↑ | 0% | 0% | 2%↑ | 1% | 0%↓ |
| Scientists | 1% | 1% | 0% | 1% | 1% | 1% | 0% | 1% | 1% | 0% |
| Online research | 1% | 0% | 1% | 1% | 2%↑ | 1% | 0% | 2%↑ | 1% | 1% |
| The media (e.g. television, radio, newspapers) | 1% | 1% | 1% | 2% | 1% | 1% | 3% | 1% | 1% | 2% |
| Other | 3% | 0% | 2% | 2% | 4% | 5%↑ | 6% | 3% | 3% | 2% |
| Does not apply to me | 5% | 4% | 6% | 3%↓ | 6% | 5% | 0% | 4% | 5% | 5% |
| None of the above | 6% | 5% | 6% | 6% | 6% | 4% | 15%↑ | 5% | 5% | 6% |
| Refusal | 1% | 0% | 0%↓ | 0%↓ | 2%↑ | 1% | 0% | 1% | 0% | 1% |
Because respondents were able to give multiple answers, total mentions may exceed 100%.
Four out of ten respondents (40%) say that someone advised them to get the flu vaccine within the last 12 months (Image 8).
Image 8. Flu Vaccine Promoters (Q23A)
Within the last 12 months, did anyone advise you to get the flu vaccine?
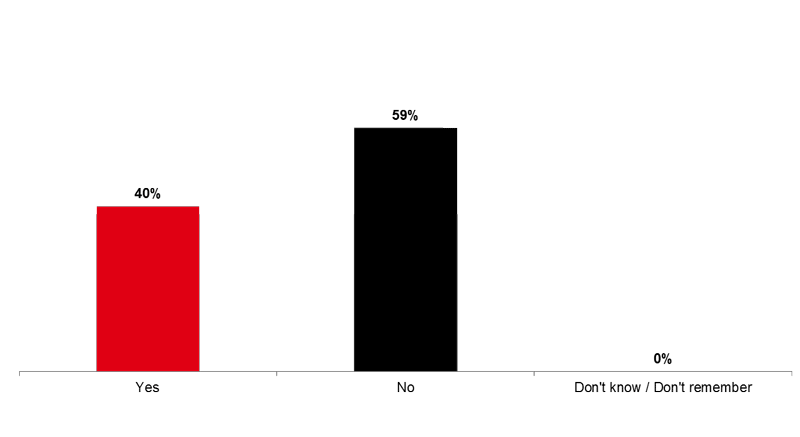
Fourteen percent of respondents (14%) say that someone advised them NOT to get the flu vaccine within the last 12 months (Image 9).
Image 9. Flu Vaccine Detractors (Q24A)
Within the last 12 months, did anyone advise you NOT to get the flu vaccine?
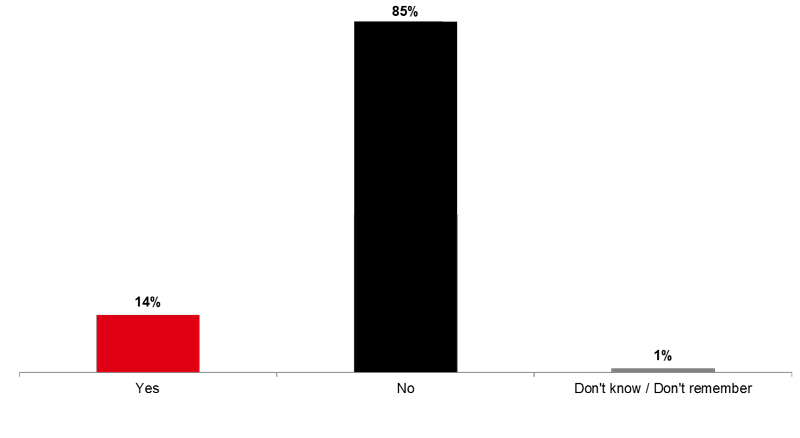
Nearly four out of ten of those who were advised to get the flu vaccine within the past year (38%) were advised by their family doctor. This proportion is higher in Ontario (44%).
Family members also play a role in advising two out of ten respondents (21%) to get the flu vaccine. To a slightly lesser extent, respondents also received this advice from their coworkers (17%) and friends (11%).
| Base: Among those who were advised to get the flu vaccine (n=787) | Total | Region | |||||
|---|---|---|---|---|---|---|---|
| Maritimes | Quebec | Ontario | MB/SK/AB | BC | Territories | ||
|
Table 15a footnotes
| |||||||
| Weighted n= | 814 | 65 | 158 | 321 | 152 | 114 | 3 |
| Unweighted n= | 787 | 76 | 173 | 251 | 155 | 117 | 15Table 15a footnote * |
| Family doctor | 38% | 45% | 31% | 44%↑ | 36% | 27%↓ | 32% |
| Family | 21% | 18% | 19% | 21% | 23% | 21% | 26% |
| Coworkers / employer | 17% | 16% | 21% | 15% | 10%↓ | 28%↑ | 12% |
| Friends | 11% | 3%↓ | 12% | 12% | 10% | 11% | 21% |
| Nurse practitioner | 6% | 11% | 9% | 3%↓ | 10%↑ | 6% | 18% |
| Pharmacist | 5% | 7% | 4% | 4% | 9%↑ | 3% | 10% |
| Medical specialist (e.g. gynecologist, cardiologist, ophthalmologist, allergist, orthopedist, psychiatrist) | 5% | 5% | 5% | 6% | 4% | 3% | 6% |
| Advertisement (e.g. media, billboards) | 4% | 2% | 3% | 4% | 5% | 2% | 8% |
| At school | 3% | 0% | 3% | 4% | 5% | 0%↓ | 0% |
| Other | 3% | 2% | 2% | 2% | 5% | 9%↑ | 14% |
| None of the above | 1% | 1% | 0% | 1% | 0% | 0% | 0% |
| Refusal | 0% | 1% | 0% | 0% | 1% | 1% | 0% |
Note: Only answers with at least 1% of total mentions are presented.
Because respondents were able to give multiple answers, total mentions may exceed 100%.
The family doctor (48%) played an important role in advising those who received the flu vaccine this year. This is consistent with the results among vulnerable groups of those ages 65+ (66%), children 6 months to 5 years (46%) and those ages 18-64 with a chronic health condition (43%).
| Total | Subgroups | Flu vaccine 2016-2017 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 18-64 | 18-64 with chronic health condition | 18-64 without chronic health condition | 65 + | Children 13-17 years of age | Children 6-12 years of age | Children 6 months to 5 years of age | Children younger than 6 months | Yes | No | ||
|
Table 15b footnotes
| |||||||||||
| Weighted n= | 814 | 678 | 195 | 482 | 136 | 89 | 128 | 107 | 9 | 352 | 462 |
| Unweighted n= | 787 | 581 | 182 | 399 | 206 | 79 | 106 | 83 | 9Table 15b footnote * | 379 | 408 |
| Family doctor | 38% | 32%↓ | 43%↑ | 28%↓ | 66%↑ | 29% | 31% | 46%↑ | 44% | 48%↑ | 30%↓ |
| Family | 21% | 22% | 13%↓ | 25%↑ | 17% | 19% | 21% | 22% | 47% | 13%↓ | 27%↑ |
| Coworkers / employer | 17% | 20% | 18% | 21% | 1%↓ | 26% | 25% | 14% | 0% | 22%↑ | 13%↓ |
| Friends | 11% | 11% | 13% | 10% | 10% | 8% | 6% | 4% | 0% | 4%↓ | 16%↑ |
| Nurse practitioner | 6% | 7% | 8% | 6% | 5% | 5% | 7% | 13%↑ | 0% | 7% | 6% |
| Pharmacist | 5% | 6% | 8% | 5% | 3% | 3% | 8% | 4% | 0% | 4% | 6% |
| Medical specialist (e.g. gynecologist, cardiologist, ophthalmologist, allergist, orthopedist, psychiatrist) | 5% | 5% | 10%↑ | 3% | 4% | 3% | 4% | 5% | 7% | 7%↑ | 3%↓ |
| Advertisement (e.g. media, billboards) | 4% | 4% | 3% | 5% | 1%↓ | 10%↑ | 8% | 4% | 3% | 1%↓ | 5%↑ |
| At school | 3% | 3% | 0%↓ | 5%↑ | 0%↓ | 0% | 2% | 0% | 0% | 2% | 3% |
| Other | 3% | 3% | 3% | 4% | 3% | 5% | 1% | 1% | 0% | 3% | 4% |
| None of the above | 1% | 0% | 0% | 1% | 1% | 2% | 2% | 1% | 0% | 1% | 1% |
| Refusal | 0% | 1% | 0% | 1% | 0% | 1% | 1% | 0% | 0% | 0% | 1% |
Note: Only answers with at least 1% of total mentions are presented.
Because respondents were able to give multiple answers, total mentions may exceed 100%.
Conversely, friends (50%) and family members (30%) played a major role in advising respondents not to get the flu vaccine.
Image 10. Flu Vaccine Detractors (Q24)
Who advised you NOT to get the flu vaccine?
Base: Among those who were advised NOT to get the flu vaccine (n=266)
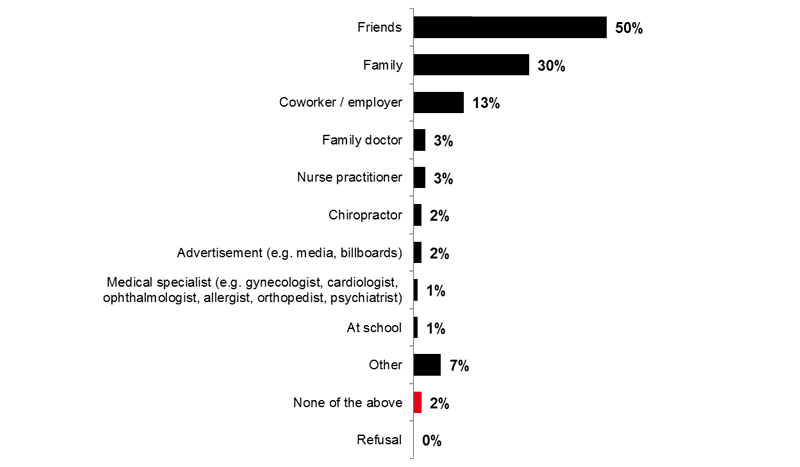
Note: Only answers with at least 1% of total mentions are presented.
Because respondents were able to give multiple answers, total mentions may exceed 100%.
More than three-quarters of respondents (78%) say that they have seen, read or heard messages promoting the flu vaccine in the last 12 months. Respondents from Manitoba, Saskatchewan and Alberta (84%) and Ontario (80%) are proportionally more likely to have seen, read or heard messages about the flu vaccine. The analysis of the results shows that there are no significant differences between the subgroups studied by Public Health Agency of Canada (PHAC).
| Total | Region | ||||||
|---|---|---|---|---|---|---|---|
| Maritimes | Quebec | Ontario | MB/SK/AB | BC | Territories | ||
| Weighted n= | 2,024 | 141 | 479 | 777 | 355 | 266 | 6 |
| Unweighted n= | 2,024 | 160 | 526 | 640 | 388 | 280 | 30 |
| Yes | 78% | 80% | 70%↓ | 80%↑ | 84%↑ | 74% | 74% |
| No | 21% | 18% | 29%↑ | 18%↓ | 16%↓ | 23% | 26% |
| Don't know / Don't remember | 2% | 3% | 1% | 1% | 1% | 3%↑ | 0% |
More than six out of ten of those who have seen, read or heard messages promoting the flu vaccine (62%) have heard them on the radio, or seen TV ads. More than one-quarter of respondents have seen messages about the flu vaccine when visiting the pharmacy, doctor's office or hospital (27%) and 16 percent have seen them on printed posters. Finally, 15 percent of respondents have seen advertisements in newspapers and 13 percent on the Internet.
The older generation has the highest proportion of those who heard messages about the flu shot on the radio or TV ads (73% of those ages 55+) and in newspapers (25%). Those 18 to 34 years of age are more likely to have seen, read or heard messages on printed posters (22%), the Internet (19%), in public transportation (6%), on Twitter or other social media (6%) and through word of mouth (5%).
Respondents from the Maritimes (37%), British Columbia (34%) and Ontario (32%) are slightly more likely to have seen, read or heard messages about the flu vaccine when visiting the pharmacy, doctor's office or hospital. On the other hand, respondents from Manitoba, Saskatchewan and Alberta (22%) are proportionally more likely to have seen messages about the flu shot on printed posters.
| Base: Among those who have seen, read or heard messages promoting the flu vaccine (n=1,569) | Total | Region | Age | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Maritimes | Quebec | Ontario | MB/SK/AB | BC | Territories | 18-34 | 35-54 | 55+ | ||
| Weighted n= | 1,57 | 112 | 337 | 624 | 297 | 196 | 4 | 416 | 608 | 546 |
| Unweighted n= | 1,569 | 125 | 375 | 514 | 320 | 212 | 23* | 255 | 532 | 782 |
| Radio / TV ads / advertisements | 62% | 58% | 63% | 65%↑ | 64% | 50%↓ | 56% | 43%↓ | 66%↑ | 73%↑ |
| When visiting the pharmacy, doctor's office, hospital | 27% | 37%↑ | 12%↓ | 32%↑ | 28% | 34%↑ | 17% | 31% | 29% | 23%↓ |
| On printed posters | 16% | 16% | 5%↓ | 18% | 22%↑ | 18% | 31% | 22%↑ | 16% | 10%↓ |
| In newspapers (articles, public health announcements or advertisements) | 15% | 13% | 17% | 13%↓ | 19%↑ | 14% | 31% | 4%↓ | 14% | 25%↑ |
| On the Internet (e.g. Facebook, PHAC website) | 13% | 13% | 14% | 13% | 14% | 12% | 15% | 19%↑ | 14% | 8%↓ |
| At workplace | 5% | 6% | 6% | 3%↓ | 5% | 9%↑ | 4% | 6% | 7%↑ | 2%↓ |
| Advertisements in public transport | 4% | 0%↓ | 0%↓ | 5%↑ | 4% | 5% | 9% | 6%↑ | 4% | 1%↓ |
| In brochures | 3% | 4% | 3% | 3% | 5% | 3% | 16% | 3% | 3% | 4% |
| Twitter or other social media | 3% | 4% | 1%↓ | 2% | 4% | 5%↑ | 4% | 6%↑ | 2% | 1%↓ |
| In magazines | 2% | 4% | 2% | 3% | 3% | 1% | 5% | 1% | 2% | 4%↑ |
| Word of mouth | 2% | 2% | 4%↑ | 2% | 3% | 1% | 6% | 5%↑ | 2% | 0%↓ |
| Other | 1% | 3%↑ | 1% | 1% | 1% | 1% | 4% | 3%↑ | 1% | 1%↓ |
| I did not see, read or heard any messaging about the flu vaccine | 1% | 1% | 1% | 1% | 1% | 1% | 0% | 1% | 1% | 1% |
| Refusal | 0% | 1% | 0% | 0%↓ | 1% | 2%↑ | 0% | 0% | 1% | 0% |
Because respondents were able to give multiple answers, total mentions may exceed 100%.
The quantitative research was conducted by telephone, using Computer Assisted Telephone Interviewing (CATI) technology. This approach is the most appropriate to assess seasonal influenza immunization coverage among different subgroups of the Canadian population, while ensuring a high level of representativeness. To achieve data reliability in all subgroups, a sample of 2,024 adult Canadians were surveyed. Only one adult respondent was surveyed within each household.
Fieldwork for the survey was conducted from February 14 to March 5, 2017. The national response rate for the survey was 20.3%. Complete call dispositions are presented in Appendix A. A pre-test of 30 interviews, 15 in each official language, was completed on February 14, 2017 (in English) and on February 16, 2017 (in French). Survey interviews lasted 7 minutes on average.
Telephone interviewing was conducted using Leger's Computer-Assisted Telephone Interviewing (CATI) technology. Leger's CATI system handles sampling electronically, randomly selecting and dialing the phone number to call. To ensure perfect coverage of a population, the sample consisted of residential phone numbers located in all Canadian provinces and territories, as well as cell phone numbers of Canadians who do not have a residential landline phone number (i.e. pre-validated cell-only numbers). Based on the Communications Monitoring Report 2016 published by the CRTC in 2016, which revealed that 23.7% of Canadian households were using a cell phone exclusively, Leger made sure that 23.7% of the final sample was derived from cell-only numbers.
Landline phone numbers were generated and cell-only numbers were purchased according to a stratified regional sampling approach. Regional quotas were applied to ensure that a sufficient number of interviews within each region of Canada and within each type of phone number (residential (1,527) and cell phone (497)). In addition to these regional quotas, fieldwork was conducted to ensure a good distribution of respondents in terms of gender (men and women) and language (English and French-speaking Canadians), using soft quotas.
A total of 2,024 adult Canadians were interviewed in all regions of the country. The national margin of error for the survey is +/- 2.18%, 19 times out of 20.
Based on data from Statistics Canada's 2011 national census, Leger weighted the results of this survey by age, gender, region and language (mother tongue). Results were also weighted based on the presence of minor children in the household (yes or no), since this question was asked and is important for this study. Moreover, the weight of respondents reached via the cell-only sample was also controlled to match the expected 23.7% after weighting.
Telephone interviewing was conducted using Leger's CATI technology. Highly trained data analysts program each survey in CATI then perform thorough testing to ensure accuracy in set-up and data collection.
Leger's CATI system handles sampling and questionnaire completion electronically, removing the possibility of misdials and imposing control over skip patterns, branching, and valid ranges. The system can be used to automate many calculations that would slow unaided interviewers.
CATI also removes the need for separate coding and data entry cycles, further reducing the opportunity for error. CATI also ensures correct timing of call-backs. To keep complaints to a minimum, no number is called twice in a two-hour period and each number is called on different days of the week and at different times of the day (i.e. late afternoon, evening, daytime). This system ensures all scheduled appointments are kept, maximizing the response rate and sample representativeness.
Leger's call centre is located in Montreal and has a total of 100 stations. All interviews for this research were conducted out of our Montreal call centre. This call centre is divided into three distinct divisions: One consisting of English only interviewers, one of French only interviewers and one of bilingual interviewers. This ensures that all telephone surveys can easily be conducted in either official language. If at any time, a potential respondent wished to conduct the survey in French (or vice-versa), they were quickly transferred to a French or bilingual interviewer, no matter when or where the call was made.
Leger integrates all projects to a Virtual Call Centre™ (VCC) platform. The Virtual Call Centre™ is a system enabling interviewers in our call centre to conduct telephone surveys supported by questionnaire and sample management provided over the Internet. The efficiency of the VCC greatly benefits the success of any project.
The VCC allows both the bilingual and English (or French) interviewers to have access to the same survey as well as the same sample data base. This is particularly important for a telephone survey that demands tight fielding timeline. We utilized all languages seamlessly to ensure that data collection was finished in the prescribed timeline. Additionally, having the VCC system allowed us to manage provincial and other quotas simultaneously.
Once the questionnaire was installed, a close validation of the programmed questionnaire was carried out to avoid any potential data error. This validation ensured that the data entry process conformed to the survey's basic logic.
The VCC system allowed for questionnaire changes to be completed quickly and accurately. By allowing the Research Manager access from anywhere in the world, Leger's research staff can receive changes and update the survey program within a very short time. This procedure allowed for the nimble management process required for the project.
Leger's call centre enables data collection supervisors to monitor interviews as they are being conducted. Monitoring is essential to ensure the proper administration of a questionnaire. Through ongoing monitoring, supervisors ensured the questionnaire was administered properly and provided timely feedback to interviewers regarding potential issues with coding or wording.
Leger's monitoring and recording system allows project managers and clients to monitor interviews as they happen. Upon request, interview recordings could be available through a secure FTP site so that clients could listen to the quality of our interviews.
Senior supervisors monitored interviews throughout field to ensure flow, clarity, consistency and comprehension of the survey. The Project Authority would have been notified immediately of any issue. If a serious issue had been found, fielding would have ceased until the problem was rectified.
With Centralus, our proprietary software, project managers could check on frequencies, the number of completes, quotas (if any), and the response rate at any time during field. This "real-time" management tool allowed for any issues to be quickly flagged and corrected.
All interviewers used by Leger during this project received customized training with respect to interview techniques, and in-depth training on our call centre CATI software. Once they have completed their probationary period, Leger's interviewers are evaluated every six months based on the following:
For each project, our field staff members go through a practice session before fielding. During these practice sessions, interviewers will work with one other to role-play a simulated interviewer/respondent survey. The field supervisor will allow interviewing to commence only when satisfied that each interviewer is able to conduct a proper, professional interview. Intensive monitoring, editing, and verification are conducted throughout the fielding process.
Phone interviews were monitored throughout the data collection period, with a minimum of 10% of each interviewer's phone calls being monitored. By insisting on ongoing monitoring, we are able to ensure the quality of the data collected.
Upon completion of data collection, Leger's data analysts and data processing department cleaned the data thoroughly, ensuring that:
The data was checked and cleaned after the first night of field and at project completion. During analysis, all numbers were double-checked and any outliers are double-checked to ensure the data has been entered accurately in the first place. If necessary, the original phone call could be reviewed to check the answer.
Low response rate threatens a survey's reliability and validity. Through Leger's experience surveying various populations, we have established the following methods to maximize response rates:
The overall response rate for this study is 20.3%.
The response rate is calculated using the following formula: (Completed interviews + Out of Sample) / (Total sample - Invalid sample). This is the Market Research and Intelligence Association's standard calculation method for the response rate of a telephone survey. The table below presents the calculation details.
| TOTAL SAMPLE | 18,545 |
|---|---|
| Invalid sample | 3,545 |
| No service | 3,361 |
| Non-residential | 65 |
| Fax / Modem / Pager | 119 |
| Numbers outside of sample | 1,023 |
| Language Barrier | 233 |
| Unqualified (deaf-mute, etc.) | 228 |
| Quota attained | 562 |
| EFFECTIVE SAMPLE | 13,977 |
| Non-completed interviews | 11,953 |
| Refusal | 4,236 |
| No answer | 2,726 |
| Answering machine | 4,159 |
| Line busy | 108 |
| Incomplete | 120 |
| Appointment | 604 |
| COMPLETED INTERVIEWS | 2,024 |
| Response rate: | 20.3% |
An effective response rate of 20.3% is average for a national telephone survey of 2,024 respondents conducted in about three weeks, matching typical rates for similar studies. This response rate minimizes the risks of an important non-response bias in the survey as it decreases the likelihood of an unrepresentative sample.
Nevertheless, a basic comparison of the unweighted and weighted sample sizes was conducted to identify potential non-response bias that could be introduced by lower response rates among specific demographic subgroups (see table below). As is typically the case for a telephone survey mainly targeting homes with a fixed telephone line, younger individuals are more difficult to reach. To compensate for this fact, Leger conducted over 497 interviews who are wireless only households (reached on their cellular phone). Using this procedure, we see that our unweighted sample closely matches the weighted numbers, devised using Statistics Canada updated data.
The table below presents the geographic distribution of respondents, before and after weighting. There were almost no imbalances in geographical distribution in the unweighted sample. The weighting process has mainly adjusted the weight of Ontario which had been slightly under-represented in the sample in order to allow more respondents in small provinces and territories.
| Unweighted | Weighted | |
|---|---|---|
| Newfoundland and Labrador | 1% | 1% |
| Prince-Edward-Island | 1% | 0% |
| Nova Scotia | 4% | 3% |
| New Brunswick | 3% | 2% |
| Quebec | 26% | 24% |
| Ontario | 32% | 38% |
| Manitoba | 4% | 4% |
| Saskatchewan | 4% | 3% |
| Alberta | 11% | 11% |
| British Columbia | 14% | 13% |
| Nunavut | 0% | 0% |
| Northwest Territories | 0% | 0% |
| Yukon | 0% | 0% |
The following tables present the demographic distribution of respondents, according to gender, age, language (mother tongue), education and household income.
First, regarding gender, we can see that weighting has adjusted the proportion of female respondents vs. male respondents, women always being slightly overrepresented in telephone surveys.
| GENDER | Unweighted | Weighted |
|---|---|---|
| Male | 40% | 48% |
| Female | 60% | 52% |
Regarding age distribution, the unweighted sample greatly exceeded the expectations of the Public Health Agency of Canada, requiring at least 50% of the sample to be 18 to 64 years of age. In fact, 72% of our unweighted sample was under 65 years of age, and 50% of the unweighted sample was under 55 years of age.
| AGE | Unweighted | Weighted |
|---|---|---|
| Between 18 and 24 | 4% | 9% |
| Between 25 and 34 | 13% | 18% |
| Between 35 and 44 | 15% | 17% |
| Between 45 and 54 | 18% | 20% |
| Between 55 and 64 | 22% | 17% |
| Between 65 and 74 | 18% | 12% |
| 75 or older | 11% | 7% |
Minor imbalances on language distribution were corrected with weighting, as presented below.
| LANGUAGE (MOTHER TONGUE) | Unweighted | Weighted |
|---|---|---|
| French | 25% | 21% |
| English | 59% | 57% |
| Other | 13% | 18% |
| French and English | 1% | 1% |
| French and other | 0% | 0% |
| English and other | 0% | 1% |
| Other and other | 1% | 2% |
The last tables present distribution based on education and household income, although those variables were not included in the weighting procedures.
| EDUCATION | Unweighted | Weighted |
|---|---|---|
| Grade 8 or less | 2% | 2% |
| Some high school | 8% | 6% |
| High School diploma or equivalent | 22% | 21% |
| Registered Apprenticeship or other trades certificate or diploma | 6% | 6% |
| College, CEGEP or other non-university certificate or diploma | 26% | 27% |
| University certificate or diploma below bachelor's level | 6% | 5% |
| Bachelor's degree | 17% | 19% |
| Post graduate degree above bachelor's level | 12% | 13% |
| Refusal | 1% | 1% |
| HOUSEHOLD INCOME | Unweighted | Weighted |
|---|---|---|
| $19,999 or less | 7% | 6% |
| between $20,000 and $39,999 | 13% | 12% |
| between $40,000 and $59,999 | 16% | 15% |
| between $60,000 and $79,999 | 12% | 13% |
| between $80,000 and $99,999 | 10% | 11% |
| between $100,000 to $149,999 | 12% | 14% |
| $150,000 and above | 10% | 12% |
| Refusal | 20% | 19% |
However, there is no evidence from the data that having achieved a different age or gender distribution prior to weighting would have significantly changed the results for this study. The relatively small sizes of weights and of the differences in responses between various subgroups suggest that data quality was not affected. The weight that was applied corrected the initial imbalance for data analysis purposes and no further manipulations were necessary.
As with all research conducted by Leger, contact information was kept entirely confidential and all information that could allow for the identification of participants was removed from the data, in accordance with the Privacy Act of Canada.
English Questionnaire
Section info
Page info
[ASK ALL]
QINF#
Good morning (afternoon, evening), my name is XX and I'm calling from LEGER research.
We are currently conducting a study on flu vaccine. This survey is led by the Public Health Agency of Canada. Your answers will help improve services that impact Canadians like you. We would therefore greatly appreciate your cooperation.
Your participation is voluntary and completely confidential. Your answers will remain anonymous and cannot affect in any way your dealings with the government of Canada.
Can I ask you a few questions? It will take no more than 10 minutes.
IF ASKED: Your opinion counts. Leger research is a renowned company throughout Canada. Today's study is about various current events and trends in society. There are no wrong answers. When may I contact you again? When would be a good time to contact you? Whom should I ask to speak with when I call back? Is there another person in your household with whom we could talk?
NOTE: If a respondent requests to speak with a study leader at Health Canada, please take his / her name and phone number and mention that a supervisor will call back to establish the link with Health Canada.
Contact:
Shelley Shackleton
Health Canada / Government of Canada
Phone number: 613-866-4093
Note to the interviewer: If a respondent asks you about the legitimacy of this project or if the respondent wants to make a complaint or a comment about this project, you must invite him/her to visit the MRIA Website: www.surveyverification.ca (English) or www.verificationsondage.ca (French), and you must give him the MRIA Project Registration Number: 20170208-382F
Section info
[ASK ALL]
QSCTDEMO1
To begin, I have a few questions about you.
Simple mention question
[ASK IF ECHA=CELL]
[SIMPLE MENTION]
QFLT1
INTERVIEWER INSTRUCTION: (DO NOT READ LIST. ONLY ONE MENTION)
| Response | Value | Attribute | Termination |
|---|---|---|---|
| Yes | 1 | n/a | RECODE LANDLINE |
| No | 2 | n/a | n/a |
| (DO NOT READ) Refusal | 9 | n/a | TERMINATE |
Question NUMERIC
[ASK ALL]
[NUMERIC: Min=18, Max=150]
[DECIMAL: 0]
[PROGRAMMER NOTES: Under 18 = terminate]
[VALIDATION: AGE]
AGE
INTERVIEWER INSTRUCTION: (ENTER THE NUMBER.)
| Response | Value | Attribute | Termination |
|---|---|---|---|
| Enter number | XXX | n/a | n/a |
| (DO NOT READ) Refusal | 999 | n/a | n/a |
CALCULATION AGEX
[PROGRAMMER NOTES: CALCULATION FROM AGE]
[VALIDATION:]
AGEX
| Response | Value | Attribute | Termination |
|---|---|---|---|
| Under 18 | 0 | n/a | TERMINATION |
| ...Between 18 and 24 | 1 | n/a | n/a |
| ...Between 25 and 34 | 2 | n/a | n/a |
| ...Between 35 and 44 | 3 | n/a | n/a |
| ...Between 45 and 54 | 4 | n/a | n/a |
| ...Between 55 and 64 | 5 | n/a | n/a |
| ...Between 65 and 74 | 6 | n/a | n/a |
| ...75 or older | 7 | n/a | n/a |
| Refusal | 999 | n/a | TERMINATION |
Simple mention question
[ASK ALL]
[SIMPLE MENTION]
LANGU
INTERVIEWER INSTRUCTION: (READ LIST. ONLY ONE MENTION)
| Response | Value | Attribute | Termination |
|---|---|---|---|
| French | 1 | n/a | n/a |
| English | 2 | n/a | n/a |
| Other | 3 | n/a | n/a |
| French and English | 7 | n/a | n/a |
| French and other | 4 | n/a | n/a |
| English and other | 5 | n/a | n/a |
| Other and other | 6 | n/a | n/a |
| (DO NOT READ) Dnk/Refusal | 9 | F | n/a |
Simple mention question
[ASK ALL]
[SIMPLE MENTION]
SEX
Enter respondent's gender - DO NOT READ.
INTERVIEWER INSTRUCTION: (DO NOT READ LIST. ONLY ONE MENTION)
| Response | Value | Attribute | Termination |
|---|---|---|---|
| Male | 1 | n/a | n/a |
| Female | 2 | n/a | n/a |
Simple mention question
[ASK ALL]
[SIMPLE MENTION]
Note: The flu vaccine can be received by a shot (needle) or nasal spray.
INTERVIEWER INSTRUCTION: (DO NOT READ LIST. ONLY ONE MENTION)
| Response | Value | Attribute | Termination |
|---|---|---|---|
| Yes | 1 | n/a | n/a |
| No | 2 | n/a | n/a |
Simple mention question
[ASK ALL]
[SIMPLE MENTION]
INTERVIEWER INSTRUCTION: (DO NOT READ LIST. ONLY ONE MENTION)
| Response | Value | Attribute | Termination |
|---|---|---|---|
| Yes | 1 | n/a | n/a |
| No | 2 | n/a | n/a |
Multiple question - open
[ASK IF Q2=2]
[MENTIONS MULTIPLES: Max=13]
[PROGRAMMER NOTES: Make sure to create a column by choice of code answers 0,1]
INTERVIEWER INSTRUCTION: DO NOT READ - MULTIPLE ANSWERS ALLOWED
Please select all that apply.
| Response | Value | Attribute | Termination |
|---|---|---|---|
| I am allergic | 01 | n/a | n/a |
| I do not like needles / I am scared of needles | 02 | n/a | n/a |
| I do not need it / I am not a person at high risk / It is not recommended for me | 03 | n/a | n/a |
| I did not have enough information | 04 | n/a | n/a |
| I did not get the time to do it | 05 | n/a | n/a |
| I had difficulty making an appointment | 06 | n/a | n/a |
| I had difficulty getting to clinic / doctor's office | 07 | n/a | n/a |
| I did not want to pay for a vaccine / too expensive | 08 | n/a | n/a |
| I did not know it existed | 09 | n/a | n/a |
| I do not believe in its effectiveness | 10 | n/a | n/a |
| I believe vaccines are unsafe (risks associated to vaccine) | 11 | n/a | n/a |
| I fear side effects | 12 | n/a | n/a |
| Other reason, please specify: | 96 | FO | n/a |
| (DO NOT READ) Refusal | 99 | XF | n/a |
| *** I was sick / I was in the hospital | 13 | N | n/a |
Simple mention question
[ASK IF Q2=1]
[SIMPLE MENTION]
INTERVIEWER INSTRUCTION: READ. ONLY ONE MENTION
| Response | Value | Attribute | Termination |
|---|---|---|---|
| Flu shot | 1 | n/a | n/a |
| Nasal spray (Flumist) | 2 | n/a | n/a |
| (DO NOT READ) Does not remember | 8 | n/a | n/a |
Simple mention question
[ASK IF Q2=1]
[SIMPLE MENTION]
[ORDER OF THE LIST: In order]
INTERVIEWER INSTRUCTION: READ. ONLY ONE MENTION
| Response | Value | Attribute | Termination |
|---|---|---|---|
| September 2016 | 0 | n/a | n/a |
| October 2016 | 1 | n/a | n/a |
| November 2016 | 2 | n/a | n/a |
| December 2016 | 3 | n/a | n/a |
| January 2017 | 4 | n/a | n/a |
| February 2017 | 5 | n/a | n/a |
| (DO NOT READ) Does not remember | 8 | n/a | n/a |
Multiple mentions question
[ASK if Q2=1]
[MENTIONS MULTIPLES: Max=10]
[PROGRAMMER NOTES: Make sure to create a column by choice of code answers 0,1]
INTERVIEWER INSTRUCTION: DO NOT READ - MULTIPLE ANSWERS ALLOWED
Please select all that apply.
| Response | Value | Attribute | Termination |
|---|---|---|---|
| I want to prevent infection / I do not want to get sick | 01 | n/a | n/a |
| I am at risk because of my health condition | 02 | n/a | n/a |
| I am at risk because of my age | 03 | n/a | n/a |
| I was recommended by a health care professional | 04 | n/a | n/a |
| I was encouraged by family members, colleagues or friends | 05 | n/a | n/a |
| It is required in my workplace | 06 | n/a | n/a |
| I would be required to wear a mask at work if not vaccinated | 07 | n/a | n/a |
| It was offered / free (by employer or other) | 08 | n/a | n/a |
| If not vaccinated, I can transmit the disease to at-risk people (children, elderly or sick people/patients) | 09 | n/a | n/a |
| If not vaccinated, I can transmit the disease to family members, colleagues or friends (without mention of at-risk people) | 10 | n/a | n/a |
| Other reason, please specify: | 96 | FO | n/a |
| (DO NOT READ) Refusal | 99 | XF | n/a |
| *** I receive it every year (no specific reason) | 11 | N | n/a |
Question simple - open
[ASK IF Q2=1]
INTERVIEWER INSTRUCTION: (READ IF NEEDED - ONE ANSWER ALLOWED)
| Response | Value | Attribute | Termination |
|---|---|---|---|
| Temporary vaccine clinic (i.e. at the mall) | 01 | n/a | n/a |
| Doctor's office / health clinic | 02 | n/a | n/a |
| CLSC / Community health centre | 03 | n/a | n/a |
| Hospital | 04 | n/a | n/a |
| Pharmacy | 05 | n/a | n/a |
| Workplace | 06 | n/a | n/a |
| Other, please specify: | 96 | FO | n/a |
| (DO NOT READ) Does not remember | 98 | XF | n/a |
| *** Retirement residence / eldercare centre | 07 | N | n/a |
Question NUMERIC
[ASK ALL]
[NUMERIC: Min=0, Max=15]
[DECIMAL: 0]
[PROGRAMMER NOTES: ALLOW UP TO 15 CHILDREN]
INTERVIEWER INSTRUCTION: We do not want the respondent to provide information about children of which he or she is not the parent or guardian, such as younger siblings. If a respondent asks if they can answer for their spouse's children, the answer would be "yes, if you are aware of the vaccines they received and their state of health"
(Record number of children:
VALIDATE THAT THEY ARE LESS THAN 18 YEARS.
IF MORE THAN 15 CHILDREN - PROVIDE A REMINDER WITH THE RESPONDENTS
| Response | Value | Attribute | Termination |
|---|---|---|---|
| None | 000 | n/a | n/a |
| (DO NOT READ) Prefer not to answer | 999 | n/a | n/a |
Numerical question
[ASK IF Q8 BIGGER THAN 0]
[NUMERIC Min=1, Max=17]
[DECIMAL: 0]
[NOTE FOR PROGRAMMING: PROGRAM FOR THE NUMBER OF CHILDREN UNDER 18 IN Q8]
[ENABLE SEIZURE IN MONTH OR YEARS]
[CAN NOT BE MORE THAN 17 YEARS]
| Response | Value | Attribute | Termination |
|---|---|---|---|
| Enter child age # 1 | Q9_1 | n/a | n/a |
| Enter child age # 2 | Q9_2 | n/a | n/a |
| Enter child age # 3 | Q9_3 | n/a | n/a |
| Enter child age # 4 | Q9_4 | n/a | n/a |
| Enter child age # 5 | Q9_5 | n/a | n/a |
| Enter child age # 6 | Q9_6 | n/a | n/a |
| Enter child age # 7 | Q9_7 | n/a | n/a |
| Enter child age # 8 | Q9_8 | n/a | n/a |
| Enter child age # 9 | Q9_9 | n/a | n/a |
| Enter child age # 10 | Q9_10 | n/a | n/a |
| Enter child age # 11 | Q9_11 | n/a | n/a |
| Enter child age # 12 | Q9_12 | n/a | n/a |
| Enter child age # 13 | Q9_13 | n/a | n/a |
| Enter child age # 14 | Q9_14 | n/a | n/a |
| Enter child age # 15 | Q9_15 | n/a | n/a |
| Response | Value | Attribute | Termination |
|---|---|---|---|
| Month(s) | 1 | n/a | n/a |
| Year(s) | 2 | n/a | n/a |
| (DO NOT READ) Refusal | 9 | n/a | n/a |
Section info
[SI Q8 = AT LEAST ONE CHILDREN]
[TO BE INCLUDED AT THE BEGINNING OF THE SECTION FOR EACH CHILD:]
[NOTES TO THE PROGRAMMER: CREATE A LOOP FOR THE NEXT SECTION FOR THE NUMBER OF CHILDREN UNDER 18 IN Q8]
LOOP ON Q10 TO Q15 * NUMBER OF CHILDREN UNDER 18 YEARS.
Let's start with your first child.
... Q10A to Q15A
Let's talk about your second child / third / child. [...]
Simple mention question
[SI Q8 = GREATER THAN 0]
[SIMPLE MENTION]
INTERVIEWER INSTRUCTION: READ. ONLY ONE MENTION
| Response | Value | Attribute | Termination |
|---|---|---|---|
| A boy | 1 | n/a | n/a |
| A girl | 2 | n/a | n/a |
| (DO NOT READ) Refusal | 9 | n/a | n/a |
Simple mention question
[ASK IF Q8 = GREATER THAN 0]
[SIMPLE MENTION]
INTERVIEWER INSTRUCTION: (DO NOT READ LIST. ONLY ONE MENTION)
| Response | Value | Attribute | Termination |
|---|---|---|---|
| Yes | 1 | n/a | n/a |
| No | 2 | n/a | n/a |
Simple mention question
[ASK IF Q9>5 MONTHS AND <10 YEARS AND Q11=1]
[SIMPLE MENTION]
INTERVIEWER INSTRUCTION: (DO NOT READ LIST. ONLY ONE MENTION)
| Response | Value | Attribute | Termination |
|---|---|---|---|
| Yes | 1 | n/a | n/a |
| No | 2 | n/a | n/a |
| (DO NOT READ) Does not know / Does not remember | 8 | n/a | n/a |
Simple mention question
[ASK IF Q9>5 MONTHS AND <10 YEARS AND Q11=1 AND Q12=1]
[SIMPLE MENTION]
INTERVIEWER INSTRUCTION: (DO NOT READ LIST. ONLY ONE MENTION)
| Response | Value | Attribute | Termination |
|---|---|---|---|
| One dose | 1 | n/a | n/a |
| Two doses | 2 | n/a | n/a |
| (DO NOT READ) Does not know / Does not remember | 8 | n/a | n/a |
Multiple mentions question
[ASK IF Q13=1]
[MENTIONS MULTIPLES: Max=12]
[PROGRAMMER NOTES: Make sure to create a column by choice of code answers 0,1]
INTERVIEWER INSTRUCTION: DO NOT READ - MULTIPLE ANSWERS ALLOWED
Please select all that apply
| Response | Value | Attribute | Termination |
|---|---|---|---|
| He/she had an allergic reaction to the first dose | 01 | n/a | n/a |
| He/she does not like needles / is scared of needles | 02 | n/a | n/a |
| He/she does not need it / It is not recommended for him/her | 03 | n/a | n/a |
| We did not have enough information | 04 | n/a | n/a |
| We did not get the time to do it | 05 | n/a | n/a |
| We had difficulty making an appointment | 06 | n/a | n/a |
| We had difficulty getting to clinic / doctor's office | 07 | n/a | n/a |
| We I did not want to pay for a second dose / too expensive | 08 | n/a | n/a |
| We did not know there was a second dose | 09 | n/a | n/a |
| I do not believe in the second dose's effectiveness | 10 | n/a | n/a |
| He/she had side effects after the first dose | 12 | n/a | n/a |
| Other reason, please specify: | 96 | FO | n/a |
| (DO NOT READ) Refusal | 99 | XF | n/a |
Multiple mentions question
[ASK IF Q11=2]
[MENTIONS MULTIPLES: Max=13]
[PROGRAMMER NOTES: Make sure to create a column by choice of code answers 0,1]
INTERVIEWER INSTRUCTION: DO NOT READ - MULTIPLE ANSWERS ALLOWED
Please select all that apply.
| Response | Value | Attribute | Termination |
|---|---|---|---|
| He/she is allergic | 01 | n/a | n/a |
| He/she does not like needles / is scared of needles | 02 | n/a | n/a |
| He/she does not need it / is not at high risk / It is not recommended for him/her | 03 | n/a | n/a |
| I did not have enough information | 04 | n/a | n/a |
| I did not get the time to do it | 05 | n/a | n/a |
| I had difficulty making an appointment | 06 | n/a | n/a |
| I had difficulty getting to clinic / doctor's office | 07 | n/a | n/a |
| I did not want to pay for a vaccine / too expensive | 08 | n/a | n/a |
| I did not know it existed | 09 | n/a | n/a |
| I do not believe in its effectiveness | 10 | n/a | n/a |
| I believe vaccines are unsafe (risks associated to vaccine) | 11 | n/a | n/a |
| I or he/she fear(s) side effects | 12 | n/a | n/a |
| Other reason, please specify: | 96 | FO | n/a |
| (DO NOT READ) Refusal | 99 | XF | n/a |
END OF LOOP - GO TO SECTION info
Now, regarding your second child / third / child.
Q10B - Q15B...
Q10C - Q15C...
FOR THE 15 CHILDREN...
Section info
HEALTH STATUS
Multiple mentions question[ASK ALL]
[MENTIONS MULTIPLES: Max=12]
[ORDER OF THE LIST: In order]
INTERVIEWER INSTRUCTION: (READ LIST - MULTIPLE ANSWERS ALLOWED)
Please select all that apply.
Check the problem only if the respondent is currently suffering or is still being treated.
| Response | Value | Attribute | Termination |
|---|---|---|---|
| Asthma | 01 | n/a | n/a |
| Another chronic lung disease such as emphysema, chronic bronchitis or cystic fibrosis | 02 | n/a | n/a |
| A heart condition such as angina, high blood pressure, heart failure, heart attack | 03 | n/a | n/a |
| Cancer | 04 | n/a | n/a |
| Diabetes or other metabolic diseases | 05 | n/a | n/a |
| Chronic liver disease | 06 | n/a | n/a |
| Chronic kidney disease | 07 | n/a | n/a |
| Immune disorder or immune suppression such as chemotherapy, radiation, steroid use or an organ transplant | 08 | n/a | n/a |
| Spleen problems or removal | 09 | n/a | n/a |
| Anemia / thalassemia/ hemoglobinpathy | 10 | n/a | n/a |
| Morbid obesity (BMI > 40) | 11 | n/a | n/a |
| Conditions that compromises management of respiratory secretions, with increased risk of aspiration | 12 | n/a | n/a |
| (DO NOT READ) None of the above | 97 | X | n/a |
| (DO NOT READ) Refusal | 99 | X | n/a |
Multiple mentions question
[ASK ALL]
[MENTIONS MULTIPLES: Max=7]
[Order of the List: Random 1 TO 6]
INTERVIEWER INSTRUCTION: (READ LIST - MULTIPLE ANSWERS ALLOWED)
Please select all that apply.
| Response | Value | Attribute | Termination |
|---|---|---|---|
| Family doctor | 01 | n/a | n/a |
| Nurse practitioner | 02 | n/a | n/a |
| Pharmacist | 03 | n/a | n/a |
| Dentist / orthodontist | 04 | n/a | n/a |
| Medical specialist (e.g. gynecologist, cardiologist, ophthalmologist, allergist, orthopedist, psychiatrist) | 05 | n/a | n/a |
| Midwife | 06 | n/a | n/a |
| Did not see a health care provider | 97 | X | n/a |
| (DO NOT READ) Refusal | 99 | X | n/a |
Multiple mentions question
[ASK ALL]
[MENTIONS MULTIPLES: Max=7]
[ORDER OF THE LIST: Random 1 to 6]
INTERVIEWER INSTRUCTION: (READ LIST. MULTIPLE ANSWERS ALLOWED)
Please select all that apply.
| Response | Value | Attribute | Termination |
|---|---|---|---|
| Chiropractor | 01 | n/a | n/a |
| Acupuncturist | 02 | n/a | n/a |
| Homeopath or naturopath | 03 | n/a | n/a |
| Herbalist | 04 | n/a | n/a |
| Reflexologist | 05 | n/a | n/a |
| Spiritual or religious healer | 06 | n/a | n/a |
| Did not see an alternative care provider | 97 | X | n/a |
| (DO NOT READ) Refusal | 99 | X | n/a |
Simple mention question
[ASK ALL]
INTERVIEWER INSTRUCTION: DO NOT READ - MULTIPLE ANSWERS ALLOWED
Please select all that apply.
| Response | Value | Attribute | Termination |
|---|---|---|---|
| Yes, please specify: | 96 | O | n/a |
| No or doesn't remember | 02 | n/a | n/a |
[ASK ALL]
[SIMPLE MENTION]
INTERVIEWER INSTRUCTION: READ. MULTIPLE ANSWERS ALLOWED
| Response | Value | Attribute | Termination |
|---|---|---|---|
| Yes, with severe symptoms (e.g. hospitalization, pneumonia) | 1 | n/a | n/a |
| Yes, with mild symptoms (e.g. sudden onset of high fever, chills, sore throat, cough, muscle pain) | 2 | n/a | n/a |
| No, did not have the flu | 3 | n/a | n/a |
| (DO NOT READ) Does not know / Does not remember | 8 | n/a | n/a |
[ASK ALL]
[SIMPLE MENTION]
INTERVIEWER INSTRUCTION: READ. MULTIPLE ANSWERS ALLOWED
| Response | Value | Attribute | Termination |
|---|---|---|---|
| Yes, with severe symptoms (e.g. hospitalization, pneumonia) | 1 | n/a | n/a |
| Yes, with mild symptoms (e.g. sudden onset of high fever, chills, sore throat, cough, muscle pain) | 2 | n/a | n/a |
| No, did not have the flu | 3 | n/a | n/a |
| (DO NOT READ) Does not know / Does not remember | 8 | n/a | n/a |
Multiple mention question
[ASK ALL]
[MENTIONS MULTIPLES]
INTERVIEWER INSTRUCTION: READ. MULTIPLE ANSWERS ALLOWED
| Response | Value | Attribute | Termination |
|---|---|---|---|
| Yes, with severe symptoms (e.g. hospitalization, pneumonia) | 1 | n/a | n/a |
| Yes, with mild symptoms (e.g. sudden onset of high fever, chills, sore throat, cough, muscle pain) | 2 | n/a | n/a |
| No, don't know anyone who had the flu | 3 | n/a | n/a |
| (DO NOT READ) Does not know / Does not remember | 8 | n/a | n/a |
Multiple mention question
[ASK ALL]
[MENTIONS MULTIPLES]
INTERVIEWER INSTRUCTION: READ. MULTIPLE ANSWERS ALLOWED
| Response | Value | Attribute | Termination |
|---|---|---|---|
| Yes, with severe symptoms (e.g. hospitalization, pneumonia) | 1 | n/a | n/a |
| Yes, with mild symptoms (e.g. sudden onset of high fever, chills, sore throat, cough, muscle pain) | 2 | n/a | n/a |
| No, don't know anyone who had the flu | 3 | n/a | n/a |
| (DO NOT READ) Does not know / Does not remember | 8 | n/a | n/a |
Section info
HEALTH PROMOTION AND MESSAGING
Multiple mentions question
[ASK ALL]
[MENTIONS MULTIPLES: Max=6]
[ORDER OF THE LIST: Random 1 to 5]
INTERVIEWER INSTRUCTION: (DO NOT READ LIST. MULTIPLE ANSWERS ALLOWED)
Please select all that apply.
| Response | Value | Attribute | Termination |
|---|---|---|---|
| A public health setting, health clinic or pharmacy | 01 | n/a | n/a |
| A government website (e.g. Public Health Agency of Canada) | 02 | n/a | n/a |
| The media (e.g. newspaper, radio) | 03 | n/a | n/a |
| Social media (e.g. Twitter, Facebook) | 04 | n/a | n/a |
| An app (e.g. ImmunizeCA) | 05 | n/a | n/a |
| Other; specify | 06 | O | n/a |
| Does not apply to me | 07 | X | n/a |
| (DO NOT READ) None of the above | 97 | X | n/a |
| (DO NOT READ) Refusal | 99 | X | n/a |
Multiple mentions question
[ASK ALL]
[MULTIPLE ANSWERS: Max=6]
[ORDER OF THE LIST: Random 1 to 5]
INTERVIEWER INSTRUCTION:(READ LIST. MULTIPLE ANSWERS ALLOWED)
Please select all that apply.
| Response | Value | Attribute | Termination |
|---|---|---|---|
| Family doctor | 01 | n/a | n/a |
| Other health care provider (e.g. nurse, pharmacist, medical specialist) | 02 | N | n/a |
| Nurse practitioner | 06 | n/a | n/a |
| Pharmacist | 08 | n/a | n/a |
| Medical specialist (e.g. gynecologist, cardiologist, ophthalmologist, allergist, orthopedist, psychiatrist) | 09 | n/a | n/a |
| An alternative care provider (e.g. naturopath, homeopath) | 03 | n/a | n/a |
| Friends | 04 | n/a | n/a |
| Family | 05 | n/a | n/a |
| Other, specify | 96 | O | n/a |
| Does not apply to me | 07 | X | n/a |
| (DO NOT READ) None of the above | 97 | X | n/a |
| (DO NOT READ) Refusal | 99 | X | n/a |
Simple mention question
[ASK ALL]
[SIMPLE MENTION]
INTERVIEWER INSTRUCTION: (DO NOT READ LIST. ONLY ONE MENTION)
| Response | Value | Attribute | Termination |
|---|---|---|---|
| Yes | 1 | n/a | n/a |
| No | 2 | n/a | n/a |
| (DO NOT READ) Does not know / Does not remember | 8 | n/a | n/a |
Multiple mentions question
[ASK IF Q23A=1]
[MULTIPLE ANSWERS: Max=12]
[ORDER OF THE LIST: Random 1 to 5]
INTERVIEWER INSTRUCTION: (DO NOT READ LIST. MULTIPLE ANSWERS ALLOWED)
Please select all that apply.
| Response | Value | Attribute | Termination |
|---|---|---|---|
| Family doctor | 01 | n/a | n/a |
| Nurse practitioner | 02 | n/a | n/a |
| Pharmacist | 03 | n/a | n/a |
| Medical specialist (e.g. gynecologist, cardiologist, ophthalmologist, allergist, orthopedist, psychiatrist) | 04 | n/a | n/a |
| Midwife | 05 | n/a | n/a |
| Chiropractor | 06 | n/a | n/a |
| Acupuncturist | 07 | n/a | n/a |
| Homeopath or naturopath | 08 | n/a | n/a |
| Herbalist | 09 | n/a | n/a |
| Reflexologist | 10 | n/a | n/a |
| Spiritual or religious healer | 11 | n/a | n/a |
| Friends | 12 | n/a | n/a |
| Family | 13 | n/a | n/a |
| Other (please specify) | 96 | O | n/a |
| (DO NOT READ OUT) None of the above | 97 | X | n/a |
| (DO NOT READ OUT) Refusal | 99 | X | n/a |
Simple mention question
[ASK ALL]
[SIMPLE MENTION]
INTERVIEWER INSTRUCTION: (DO NOT READ LIST. ONLY ONE MENTION)
| Response | Value | Attribute | Termination |
|---|---|---|---|
| Yes | 1 | n/a | n/a |
| No | 2 | n/a | n/a |
| (DO NOT READ) Does not know / Does not remember | 8 | n/a | n/a |
Multiple mentions question
[ASK IF Q24A=1]
[MULTIPLE ANSWERSS: Max=14]
[ORDER OF THE LIST: Random 1 to 5]
INTERVIEWER INSTRUCTION: (DO NOT READ LIST.)
Please select all that apply.
| Response | Value | Attribute | Termination |
|---|---|---|---|
| Family doctor | 01 | n/a | n/a |
| Nurse practitioner | 02 | n/a | n/a |
| Pharmacist | 03 | n/a | n/a |
| Medical specialist (e.g. gynecologist, cardiologist, ophthalmologist, allergist, orthopedist, psychiatrist) | 04 | n/a | n/a |
| Midwife | 05 | n/a | n/a |
| Chiropractor | 06 | n/a | n/a |
| Acupuncturist | 07 | n/a | n/a |
| Homeopath or naturopath | 08 | n/a | n/a |
| Herbalist | 09 | n/a | n/a |
| Reflexologist | 10 | n/a | n/a |
| Spiritual or religious healer | 11 | n/a | n/a |
| Friends | 12 | n/a | n/a |
| Family | 13 | n/a | n/a |
| Other (please specify) | 96 | O | n/a |
| (DO NOT READ OUT) None of the above | 97 | X | n/a |
| (DO NOT READ OUT) Refusal | 99 | X | n/a |
Simple mention question
[ASK ALL]
[SIMPLE MENTION]
INTERVIEWER INSTRUCTION: (DO NOT READ LIST. ONLY ONE MENTION)
| Response | Value | Attribute | Termination |
|---|---|---|---|
| Yes | 1 | n/a | n/a |
| No | 2 | n/a | n/a |
| (DO NOT READ) Does not know / Does not remember | 8 | n/a | n/a |
Multiple mentions question
[ASK IF Q25A=1]
[MENTIONS MULTIPLES: Max=12]
[ORDER OF THE LIST: Random 1 to 5]
INTERVIEWER INSTRUCTION: (DO NOT READ LIST. MULTIPLE ANSWERS ALLOWED)
Please select all that apply.
| Response | Value | Attribute | Termination |
|---|---|---|---|
| On printed posters | 01 | n/a | n/a |
| In brochures | 02 | n/a | n/a |
| In radio / TV ads / advertisements | 03 | n/a | n/a |
| In newspapers (articles, public health announcements or advertisements) | 04 | n/a | n/a |
| In magazines | 05 | n/a | n/a |
| Advertisements in public transport | 06 | n/a | n/a |
| On the Internet (eg Facebook, PHAC website) | 07 | n/a | n/a |
| Twitter or other social media | 08 | n/a | n/a |
| When visiting the pharmacy, doctor's office, hospital | 09 | n/a | n/a |
| Other (Please specify) | 96 | O | n/a |
| (DO NOT READ OUT) I did not see, read or heard any messaging about the flu vaccine | 97 | X | n/a |
| (DO NOT READ OUT) Refusal | 99 | X | n/a |
Section info
QSCTDEMO
The next questions are for statistical purposes only. It will allow us to group your answers with those of other similar respondents.
Scola
[ASK ALL]
[SIMPLE MENTION]
[ORDER OF THE LIST: In order]
INTERVIEWER INSTRUCTION: (READ LIST. ONLY ONE ANSWER)
| Response | Value | Attribute | Termination |
|---|---|---|---|
| Grade 8 or less | 1 | n/a | n/a |
| Some high school | 2 | n/a | n/a |
| High School diploma or equivalent | 3 | n/a | n/a |
| Registered Apprenticeship or other trades certificate or diploma | 4 | n/a | n/a |
| College, CEGEP or other non-university certificate or diploma | 5 | n/a | n/a |
| University certificate or diploma below bachelor's level | 6 | n/a | n/a |
| Bachelor's degree | 7 | n/a | n/a |
| Post graduate degree above bachelor's level | 8 | n/a | n/a |
| (DO NOT READ) Prefer not to answer | 9 | n/a | n/a |
Reven
[ASK ALL]
[SIMPLE MENTION]
[ORDER OF THE LIST: In order]
INTERVIEWER INSTRUCTION: (READ LIST. ONE MENTION POSSIBLE.)
| Response | Value | Attribute | Termination |
|---|---|---|---|
| ...Under $20,000 | 1 | n/a | n/a |
| ... $20,000 to $39,999 | 2 | n/a | n/a |
| ... $40,000 to $59,999 | 3 | n/a | n/a |
| ... $60,000 to $79,999 | 4 | n/a | n/a |
| ... $80,000 to $99,999 | 5 | n/a | n/a |
| ... $100,000 to $149,999 | 6 | n/a | n/a |
| ...$150,000 and above | 7 | n/a | n/a |
| (DO NOT READ) Refusal | 9 | n/a | n/a |
Simple mention question
[ASK ALL]
[SIMPLE MENTION]
INTERVIEWER INSTRUCTION: (DO NOT READ LIST. ONLY ONE MENTION)
| Response | Value | Attribute | Termination |
|---|---|---|---|
| Yes | 1 | n/a | n/a |
| No | 2 | n/a | n/a |
| (DO NOT READ) Refusal | 9 | n/a | n/a |
Simple mention - open
[ASK IF Q16=2]
INTERVIEWER INSTRUCTION: (DO NOT READ LIST. ONLY ONE MENTION)
| Response | Value | Attribute | Termination |
|---|---|---|---|
| China | 01 | n/a | n/a |
| France | 02 | n/a | n/a |
| Germany | 03 | n/a | n/a |
| Greece | 04 | n/a | n/a |
| Guyana | 05 | n/a | n/a |
| Hong Kong | 06 | n/a | n/a |
| India | 07 | n/a | n/a |
| Iran | 08 | n/a | n/a |
| Italy | 09 | n/a | n/a |
| Jamaica | 10 | n/a | n/a |
| Lebanon | 11 | n/a | n/a |
| Netherlands / Holland | 12 | n/a | n/a |
| Pakistan | 13 | n/a | n/a |
| Philippines | 14 | n/a | n/a |
| Poland | 15 | n/a | n/a |
| Portugal | 16 | n/a | n/a |
| Romania | 17 | n/a | n/a |
| South Korea | 18 | n/a | n/a |
| Sri Lanka | 19 | n/a | n/a |
| Taiwan | 20 | n/a | n/a |
| Trinidad and Tobago | 21 | n/a | n/a |
| United Kingdom | 22 | n/a | n/a |
| United States | 23 | n/a | n/a |
| Vietnam | 24 | n/a | n/a |
| Other country, please specify: | 96 | O | n/a |
| (DO NOT READ) Refusal | 99 | n/a | n/a |
| *** Algeria | 25 | N | n/a |
| *** Belgium | 26 | N | n/a |
| *** Colombia | 27 | N | n/a |
| *** Afghanistan | 28 | N | n/a |
| *** Bangladesh | 29 | N | n/a |
| *** Turkey | 30 | N | n/a |
| *** Ukraine | 31 | N | n/a |
| *** Africa | 32 | N | n/a |
Numerical question
[ASK IF Q16=2]
[NUMERIC: BORNES Min=1, Max=100]
[DECIMAL: 0]
[VALIDATION:]
INTERVIEWER INSTRUCTION: (RECORD NUMBER OF YEARS.)
| Response | Value | Attribute | Termination |
|---|---|---|---|
| Record number of years: | 000 | n/a | n/a |
| (DO NOT READ) Refusal | 999 | n/a | n/a |
Simple mention question
[ASK ALL]
[SIMPLE MENTION]
INTERVIEWER INSTRUCTION: (DO NOT READ LIST. ONLY ONE MENTION)
| Response | Value | Attribute | Termination |
|---|---|---|---|
| Yes, everyday | 1 | n/a | n/a |
| Yes, occasionally | 2 | n/a | n/a |
| No, never | 3 | n/a | n/a |
| No, past smoker | 4 | n/a | n/a |
| (DO NOT READ) Refusal | 9 | n/a | n/a |
Thank you for your participation.