|
[Table of Contents]
|
|
Canada Communicable Disease Report
- Supplement
Volume: 23S8
December 1997
INFECTION CONTROL GUIDELINES
Preventing Infections Associated with Indwelling Intravascular
Access Devices
Selection of Intravascular Access Devices
The characteristics and properties of the intravascular device materials
may be related to thrombogenicity and infectious complications. Materials
that are more irritating to the vascular endothelium and associated with
greater thrombogenicity have been shown to lead to an increased risk of
colonization and device-associated infection. Intravascular devices composed
of polyvinyl chloride and polyethylene are thought to be more thrombogenic
and associated with a higher risk of infection than devices composed of
materials such as polyurethane, Teflon®, silicone, steel,
or titanium. In vitro studies have suggested that certain microorganisms
may adhere more readily to polyvinyl chloride devices than to polyurethane
or Teflon® ones(20-23). The size and number
of lumens present have been suggested as factors that may be associated
with an increased risk of infection, but results of studies are conflicting(24-26).
The use of intravascular devices with an attached Dacron®
cuff, an attachable cuff impregnated with silver, heparin bonding, and
antimicrobial, and/or antiseptic (see Appendix
III) bonding represent newer technological advances. Such devices
have been recently evaluated and found to reduce intravascular device-associated
infections(27-30).
RECOMMENDATIONS
Indications for Use of Intravascular Devices
-
Intravascular devices should be used only for definite therapeutic
or diagnostic indications and should be discontinued as soon as possible.
(Category A; Grade III)
-
Intravascular devices that have been designated for delivery of parenteral
nutrition should not be used for other purposes(11,31).
(Category A; Grade II)
-
The simplest possible configuration (minimum number of lumens, connectors
and ports) (see Figures 2 and 3)
should be selected for the intended purpose of the line(25,26).
(Category B; Grade II)
Peripheral Venous Lines
-
Cannulae composed of polyurethane, silicone or Teflon®,
or butterfly steel needles should be chosen according to the indications
for their use(20,22). (Category B; Grade II)
-
There are no data to suggest that stainless steel cannulae are superior
or inferior to other cannula materials in preventing infection. (Category
C)
-
There are insufficient data to recommend for or against the use of
antimicrobial- or antiseptic-impregnated cannulae. (Category C)
Arterial Lines
-
Cannulae composed of polyurethane or Teflon® should
be chosen(20). (Category B; Grade II)
-
There are no data to recommend for or against the use of antimicrobial-
or antiseptic-impregnated cannulae. (Category C)
Central Venous Lines (including peripherally inserted
central catheters [PICC], central hemodialysis lines and central pulmonary
arterial catheters) and Midline Catheters
-
The cannulae with the simplest configuration (minimum number of lumens,
connectors and ports) (see Figures 2 and 3)
should be selected(24-26). (Category B; Grade II)
-
Cannulae composed of polyurethane, silicone, or Teflon®
should be selected(20,22,32). (Category B; Grade II)
-
The use of attachable silver-impregnated cuffs(29,30),
antiseptic- or antimicrobial- impregnated cannulae, or heparin-bonded
cannulae(33) may be indicated for institutions unable to
achieve benchmark targets for infection rates. (Category A; Grade
I)
- Cuffed tunnelled catheters or totally implantable devices as appropriate
for the intended purpose should be used for patients requiring long-term
vascular access(32,34-37). (Category B; Grade II)
Figure 2
Components of pressure monitoring system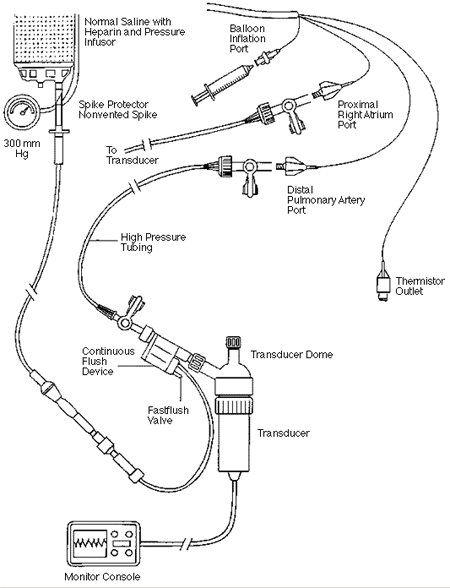
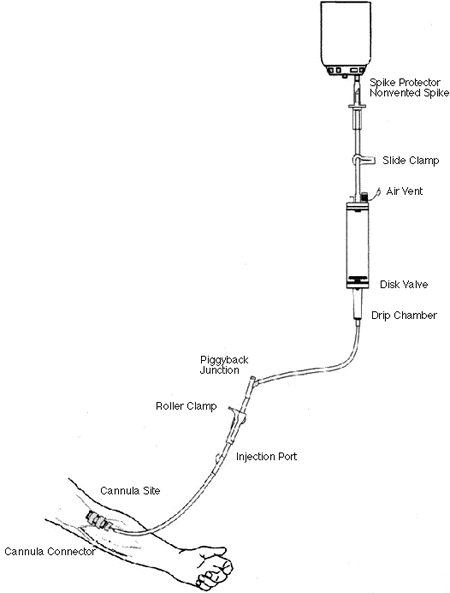
Figure 3
Diagram of IV tubing containing IV fluid roller clamp, injection port
and IV cannula
[Previous]
[Table of Contents] [Next]
|
![]()





