| |
MATERIAL AND TECHNIQUES
CT-Scan
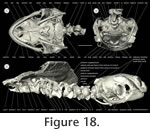
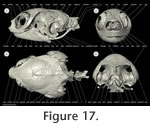 I used micro computed tomography (µ-CT) to document the cranial bones and cervical vertebrae of a subadult Emydura subglobosa specimen (PIMUZ labNo. 2009.37, CL: 63.58 mm,
Figure 17,
Figure 18). The micro-CT method is commonly used in medical studies and allows non-destructive data acquisition of anatomical structures (Morton et al. 1990;
Ritman 2004) with a high spatial I used micro computed tomography (µ-CT) to document the cranial bones and cervical vertebrae of a subadult Emydura subglobosa specimen (PIMUZ labNo. 2009.37, CL: 63.58 mm,
Figure 17,
Figure 18). The micro-CT method is commonly used in medical studies and allows non-destructive data acquisition of anatomical structures (Morton et al. 1990;
Ritman 2004) with a high spatial
 resolution. In this study, the acquisition of digital three-dimensional data was performed with a medical Scanco® micro-CT 80 (www.scanco.ch) at the Anthropological Institute of the University of Zürich. The scanning process with an isotropic voxel resolution of 0.036 mm took about 3 hours and resulted in an isq-file size of about 2 Gigabytes with a standard resolution of 1024 × 1024 pixels per slide. The 3D-software Amira 4.1.2. (Visage Imaging Richmond, Australia) was used to create a surface rendering of the data (Appendix
2, Appendix 3,
Appendix 4). resolution. In this study, the acquisition of digital three-dimensional data was performed with a medical Scanco® micro-CT 80 (www.scanco.ch) at the Anthropological Institute of the University of Zürich. The scanning process with an isotropic voxel resolution of 0.036 mm took about 3 hours and resulted in an isq-file size of about 2 Gigabytes with a standard resolution of 1024 × 1024 pixels per slide. The 3D-software Amira 4.1.2. (Visage Imaging Richmond, Australia) was used to create a surface rendering of the data (Appendix
2, Appendix 3,
Appendix 4).
 Clearing and Staining Clearing and Staining
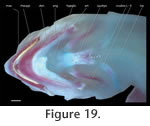
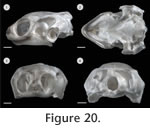
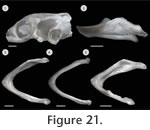
In order to study the spatial anatomy of cartilages in addition to ossified structures (Figure 19,
Figure 20,
Figure 21), one subadult specimen (PIMUZ labNo. 2009.9, CL: 79.2 mm) was cleared and double-stained following standard protocols (Taylor and Van Dyke 1985).
Serial Sections and 3D-Reconstruction
I had access to a serial section series of a hatchling specimen (collection of Prof. Dr. Wolfgang Maier, Tübingen/Germany, CL: 28.5mm), which was stained with Heidenhain's Azan (Romeis 1989) and embedded with Euparal.
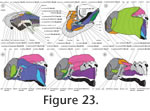
 Each slice had a thickness of 20 ?m. Every second section was digitised with a Leica DFC420 C camera mounted to a Leica M165C stereo microscope. The colour images were saved in tif format at 2592 x 1944 pixel resolution including a micrometer scale. The software Amira 4.1.2. (Visage Imaging Richmond, Australia) was used to align the images (Figure 22,
Figure 23,
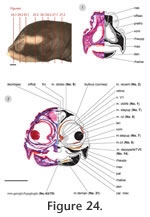
Figure 24,
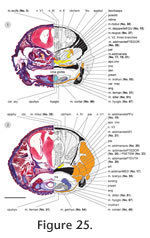
Figure 25,
Figure 26,
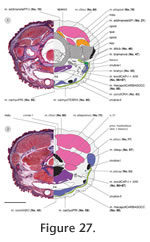
Figure 27). The technical limitations of the software and available computing power necessitated the conversion of images to greyscale mode and to 50% of the original size. Based on manually defined contour lines derived interactively from the digital images, surface renderings were provided by the software. Artefacts in the histological sections caused unevenness in the rendered surfaces of the anatomical structures. However, smoothing was avoided in order to reduce changes of the raw data, and re-drawings of the 3D reconstructions were prepared (Figure 22,
Figure 23). Each slice had a thickness of 20 ?m. Every second section was digitised with a Leica DFC420 C camera mounted to a Leica M165C stereo microscope. The colour images were saved in tif format at 2592 x 1944 pixel resolution including a micrometer scale. The software Amira 4.1.2. (Visage Imaging Richmond, Australia) was used to align the images (Figure 22,
Figure 23,
Figure 24,
Figure 25,
Figure 26,
Figure 27). The technical limitations of the software and available computing power necessitated the conversion of images to greyscale mode and to 50% of the original size. Based on manually defined contour lines derived interactively from the digital images, surface renderings were provided by the software. Artefacts in the histological sections caused unevenness in the rendered surfaces of the anatomical structures. However, smoothing was avoided in order to reduce changes of the raw data, and re-drawings of the 3D reconstructions were prepared (Figure 22,
Figure 23).
 Manual Dissection Manual Dissection
For manual dissection
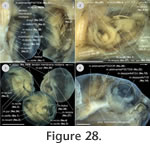
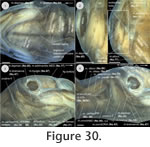
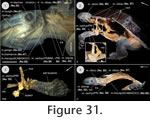
(Figure 28,
Figure 29,
Figure 30,
Figure 31), four one-year-old subadult specimens (PIMUZ labNo. 2009.37-40, CL: ranging around 65 mm) and one adult specimen (PIMUZ labNo. 2009.10, CL: 96 mm) were studied. All these specimens were obtained from the pet trade, were intoxicated and mortified in chlorobutanol, fixed in a 4% formalin solution and preserved in 70% ethanol. In the hatchling (serial section), all muscles already reached attachment sites. Though all specimens studied in this study form a postembryonic ontogenetic series, the origin and insertion sites of the muscles, which eventually changed, were accurately recorded and discussed; variability was also documented. The branching pattern of cranial nerves was checked where possible to enable homologisations of the muscular structure in addition to other criteria such as origin, insertion or the tendinuous framework (Appendix 1).
 Before manual dissection, selected specimens were stained with alizarin-red in a diluted Borax solution (Borax 1:water 5) on an automated shaker for about 12–36 h, resulting in an irreversible staining of the bones in red, while muscles, nerves, and other soft tissue elements kept their whitish colouration. However, this procedure was not as successful as previously demonstrated for teleost fishes (Hertwig
2008; Werneburg and Hertwig 2009), due to the thick skin and very dense bones of turtles when compared to non-tetrapod vertebrates. As the specimens studied were relatively fresh, a good contrast was still visible between the whitish bones and the yellowish muscles; pigment cell sprinkled muscular structures. Before manual dissection, selected specimens were stained with alizarin-red in a diluted Borax solution (Borax 1:water 5) on an automated shaker for about 12–36 h, resulting in an irreversible staining of the bones in red, while muscles, nerves, and other soft tissue elements kept their whitish colouration. However, this procedure was not as successful as previously demonstrated for teleost fishes (Hertwig
2008; Werneburg and Hertwig 2009), due to the thick skin and very dense bones of turtles when compared to non-tetrapod vertebrates. As the specimens studied were relatively fresh, a good contrast was still visible between the whitish bones and the yellowish muscles; pigment cell sprinkled muscular structures.
 Nomenclature Nomenclature
The nomenclature of the neuro- and dermatocranial elements of turtles follows mainly
Gaffney (1972,
1979). Splanchnocranial elements follow
Fürbringer (1922) for the hyal apparatus and Siebenrock (1899) for the laryngeal apparatus. Cervical vertebrae nomenclature follows
Herrel et al. (2008) (Figure 5). Shell element nomenclature follows
Zangerl (1969). Nomenclature of the brain and cranial nerves is that of
Shiino (1913).
Nomenclature of cranial musculature is newly defined, described, and depicted in
Appendix 1 and
Figure 6,
Figure 7,
Figure 8,
Figure 9,
Figure 10,
Figure 11,
Figure 12,
Figure 13,
Figure 14, and
Figure 15. Nomenclature and selected synonyms of remaining anatomical structures, such as tendinuous or overall spatial structures are listed in
Appendix 5. To create the synonym-lists for 88 cranial muscular units (Appendix 1), more than 100 original references were compared, which discuss the cranial soft tissue elements of at least 70 turtle species following the taxonomic nomenclature of
Fritz and Havaš (2007) (Appendix
6). An abbreviation guide can be found below; a list of abbreviations is presented in
Appendix 7.
|