Common menu bar links
Institutional links
Diseases & Conditions
Health & Safety
Research & Statistics
Agency Information
Search Box
E-mail this page
Canadian Integrated Program for Antimicrobial Resistance Surveillance (CIPARS)
Canadian Integrated Program for Antimicrobial Resistance Surveillance (CIPARS)
About CIPARS
- Background
- A Call for Action
- Building the System
- Overview
- Mandate
- Changes to CIPARS
- Contact Information
Background
Antimicrobial resistance (AMR) is an issue of increasing public
concern. In addition to reducing our ability to effectively
treat bacterial infections in humans and animals, AMR presents a
serious economic challenge. Although the precise financial
burden associated with AMR is not known, it is estimated that
resistance at least doubles the cost of treating a bacterial
infection and adds between $40 and $52 million per year to indirect
and direct health care costs in Canada (CCAR  , 2002).
, 2002).
Antimicrobial resistance in human medicine is primarily
associated with the use of antimicrobials to treat human infections
(WHO  , 2000; HC 2002). However,
significant amounts of antimicrobials are also used in agri-food
production, and the contamination of animals and animal products
with antimicrobial resistant bacteria has been identified as a
source for human infection with resistant organisms (JETACAR
, 2000; HC 2002). However,
significant amounts of antimicrobials are also used in agri-food
production, and the contamination of animals and animal products
with antimicrobial resistant bacteria has been identified as a
source for human infection with resistant organisms (JETACAR  1999; Poppe et
al, 1998; Wall et al, 1994; Molbak et al,
1999, Fone and Barker, 1994).
1999; Poppe et
al, 1998; Wall et al, 1994; Molbak et al,
1999, Fone and Barker, 1994).
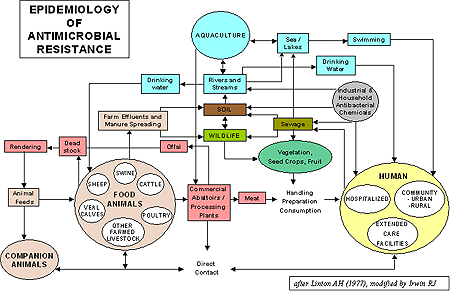
This figure describes how antimicrobials used in many sectors (circles) contribute to the emergence and spread of resistant micro-organisms in various settings (squares). Each of these are linked through direct and indirect pathways (eg. Direct pet to human contact or indirect from water or food consumption).
A Call for Action
International public health authorities are urging countries to
implement integrated AMR surveillance systems. These systems
are needed to implement sound public health interventions and to
enhance prudent use practices in human and veterinary
medicine. Surveillance is also necessary to support the
development of international food safety standards. Multiple
committees representing the joint Food and Agriculture Organization (FAO)  of the United Nations/World Health Organization (WHO)
of the United Nations/World Health Organization (WHO)  and the Codex
Alimentarius Commission
and the Codex
Alimentarius Commission  are currently
examining the issue of AMR. Food animal producers are under
increasing pressure to restrict antimicrobial use to meet domestic
and international market demands.
are currently
examining the issue of AMR. Food animal producers are under
increasing pressure to restrict antimicrobial use to meet domestic
and international market demands.
In Canada, the establishment of a national surveillance system
to monitor AMR and use in the agri-food and agriculture sectors and
the impact of resistance on human health was formally recommended
in 1997 at the national consensus conference “Controlling
Antimicrobial Resistance: An Integrated Action Plan for
Canadians” co-convened by Health Canada and the Canadian
Infectious Diseases Society. Subsequently, this
recommendation received further endorsement from several national
external advisory committees to Health Canada; the Enteric Disease
Surveillance (ENDS) Steering Committee, the Canadian Committee on
Antibiotic Resistance (CCAR) , and most recently the Advisory
Committee on Animal Uses of Antimicrobials and Impact on Resistance
and Human Health. This latter committee stated in their 2002
report that “In Canada, as in most countries, the
[surveillance] data are fragmentary, often biased, focused on a
narrow and variable range of bacterial pathogens, collected in an
unsystematic way and not generally compatible between laboratories
and/or countries because methods used for testing resistance have
not been standardized” (HC, 2002).
, and most recently the Advisory
Committee on Animal Uses of Antimicrobials and Impact on Resistance
and Human Health. This latter committee stated in their 2002
report that “In Canada, as in most countries, the
[surveillance] data are fragmentary, often biased, focused on a
narrow and variable range of bacterial pathogens, collected in an
unsystematic way and not generally compatible between laboratories
and/or countries because methods used for testing resistance have
not been standardized” (HC, 2002).